Product Images Eptifibatide
View Photos of Packaging, Labels & Appearance
Product Label Images
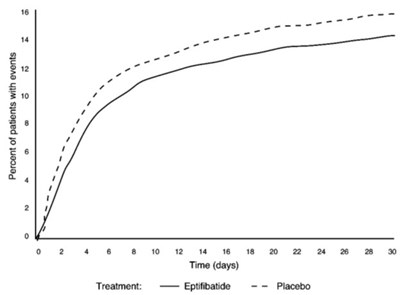
The following 5 images provide visual information about the product associated with Eptifibatide NDC 72078-025 by Mylan Institutional Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Carton 20 mg - image 03

This text is a description of Eptifibatide Injection. Each vial contains 20 mg/10 mL of eptifibatide, and the recommended dose is displayed on the accompanying package insert. The injection is for intravenous use only, and the vial should be discarded if not used within two months of transfer to 25°C. The injection is made in India and is manufactured for Mylan Institutional LLC in the USA. Novaplus is a registered trademark of Viient, Inc. There is also a code number and an NDC on the vial.*
Carton 75 mg - image 04
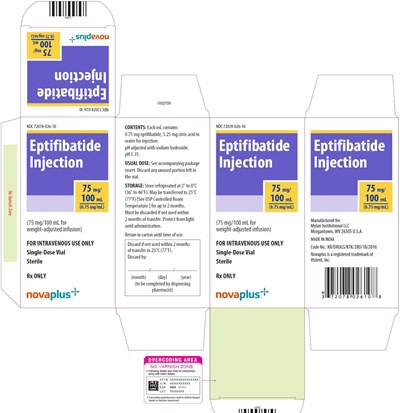
This is description of a medication named Eptifibatide Injection. It is a 75mg injection that comes in single-dose vile. Each mL contains 0.75 mg Eptifibatide and 5.25 mg citric acid in water for injection. The pH is adjusted with sodium hydroxide and is set to 5.35. The usual dose is mentioned in the package insert, and the unused portion left in the vial should be discarded. The medication must be refrigerated at 2°C to 8°C (36 to 46°F) and can be transferred to 25°C (77°F) temperature for up to two months. It must be discarded if not used within 2 months from the date of manufacture. This medication is for intravenous use only and must be administered by a licensed healthcare practitioner.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.