Product Images Atorvastatin Calcium
View Photos of Packaging, Labels & Appearance
- atorvastatin-10mg1000s - atorvastatin 10mg1000s
- atorvastatin-10mg30s - atorvastatin 10mg30s
- atorvastatin-10mg500s - atorvastatin 10mg500s
- atorvastatin-10mg90s - atorvastatin 10mg90s
- atorvastatin-20mg1000s - atorvastatin 20mg1000s
- atorvastatin-20mg30s - atorvastatin 20mg30s
- atorvastatin-20mg500s - atorvastatin 20mg500s
- atorvastatin-20mg90s - atorvastatin 20mg90s
- atorvastatin-40mg1000s - atorvastatin 40mg1000s
- atorvastatin-40mg30s - atorvastatin 40mg30s
- atorvastatin-40mg500s - atorvastatin 40mg500s
- atorvastatin-40mg90s - atorvastatin 40mg90s
- atorvastatin-80mg1000s - atorvastatin 80mg1000s
- atorvastatin-80mg30s - atorvastatin 80mg30s
- atorvastatin-80mg500s - atorvastatin 80mg500s
- atorvastatin-80mg90s - atorvastatin 80mg90s
- atorvastatin-fi1-jpg - atorvastatin fi1 jpg
- atorvastatin-figu3 - atorvastatin figu3
- atorvastatin-figure2 - atorvastatin figure2
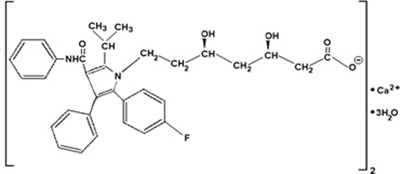
- atorvastatin-molstructure-jpg - atorvastatin molstructure jpg
Product Label Images
The following 20 images provide visual information about the product associated with Atorvastatin Calcium NDC 72134-009 by Agnitio Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
atorvastatin-10mg1000s - atorvastatin 10mg1000s

Atorvastatin Calcium Tablets, USP is a prescription medication used to lower cholesterol levels in the blood. Each film-coated tablet contains 10 mg of atorvastatin calcium, USP. It is advised to read the package insert for full prescribing information before use. The tablets should be stored at a controlled room temperature between 20-25°C (68-77°F) and dispensed in tight containers (USP). The tablets are manufactured by Halo Pharmaceutical Canada, Inc and distributed by Agnitio Inc. The NDC number for the tablets is 72134-009-04. The lot expiration is 10/2022.*
atorvastatin-10mg30s - atorvastatin 10mg30s

This is a medication called Atorvastatin Calcium Tablets, USP, containing 10mg of the active ingredient in each film-coated tablet. It is important to refer to the package insert for complete dosage and usage instructions. The medication should be stored at room temperature in tightly sealed containers. It is distributed by Agnitio Inc. with a lot number and expiration date. The manufacturer, Halo Pharmaceutical Canada, Inc, is located in Quebec, Canada. This medication can only be obtained with a prescription.*
atorvastatin-10mg500s - atorvastatin 10mg500s

Each tablet contains 10mg of atorvastatin calcium, USP, and is film-coated. The package contains 500 tablets with an expiration date of 10/2022. It is intended for prescription use and should be stored at a temperature of 20-25°C (68-77°F) in tight containers. The manufacturer is Halo Pharmaceutical Canada, Inc. Please refer to the package insert for full prescribing information.*
atorvastatin-10mg90s - atorvastatin 10mg90s

This is a medication called Atorvastatin Calcium Tablets, USP which contains 10mg of atorvastatin. It is manufactured by Halo Pharmaceutical Canada and distributed by Agnitio Inc. The medication should be stored at a controlled room temperature and dispensed in tight containers. Further information regarding the dosage and usage of this medication should be obtained from the package insert.*
atorvastatin-20mg1000s - atorvastatin 20mg1000s

This is a description of a medication called Atorvastatin Calcium Tablets, USP. Each tablet contains 20mg of atorvastatin calcium, and the recommended dosage should be followed as indicated in the package insert. It should be stored in a controlled room temperature of 20-25°C, and be dispensed in tight containers. The medication is manufactured by Halo Pharmaceutical Canada, and distributed by Agnitio Inc. The NDC number for the medication is 72134-010-04, and it is only available with a prescription. The lot number and expiration date are also indicated on the label.*
atorvastatin-20mg30s - atorvastatin 20mg30s

This is a description of Atorvastatin Calcium Tablets, USP used for prescribing information. Each film-coated tablet contains 20 mg atorvastatin calcium. The tablets should be stored at controlled room temperature (20-25°C). The product is to be kept in tight containers. The manufacturer of this product is Halo Pharmaceutical Canada, Inc. The package insert contains full dosages and use information. The NDC code for Atorvastatin Calcium Tablets, USP is 72134-010-01 with a lot expiration date of 10/2022.*
atorvastatin-20mg500s - atorvastatin 20mg500s

Atorvastatin Calcium Tablets are a prescription medication that contains 20 mg of atorvastatin calcium USP. This information advises to see the package insert for a full prescription, store the medication between 20-25 °C , dispense in tight containers, and notes the product was manufactured by Halo Pharmaceutical Canada, Inc. The NDC code is 72134-010-03 and the medication is distributed by Agnitio Inc. in Appleton, WI, USA. Lot number and expiration date are provided.*
atorvastatin-20mg90s - atorvastatin 20mg90s

This is a description of a medication containing atorvastatin calcium in a 20 mg dosage. The tablets should be stored at controlled room temperature and dispensed in tight containers. The manufacturing company is Halo Pharmaceutical Canada, Inc. and they are distributed by Agnitio Inc. The tablets come in a 90-tablet package and should be used according to the information provided in the package insert. The NDC number is 72134-010-02, and the lot number and expiration date are also provided.*
atorvastatin-40mg1000s - atorvastatin 40mg1000s
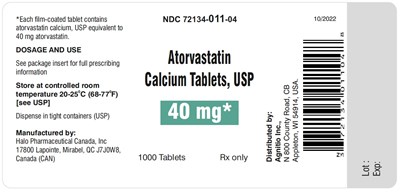
Each tablet contains 40 mg of Atorvastatin Calcium. It is used for lowering the bad cholesterol and increasing good cholesterol levels in the body. It should be stored between 20-25°C (68-77°F). The tablets should be dispensed in tight containers. The manufacturer of the medicine is Halo Pharmaceutical Canada, Inc. The text does not provide any detailed information on the usage of the medicine.*
atorvastatin-40mg30s - atorvastatin 40mg30s
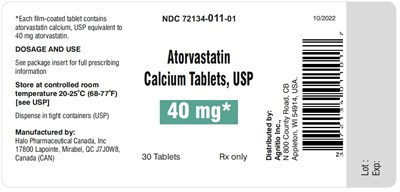
Atorvastatin Calcium Tablets, USP with 40 mg strength, sold in package containing 30 film-coated tablets. The tablets are stored at 20-25°C (68-77°F) in tight containers. For complete information on dosage and usage, see the package insert. It is manufactured by Halo Pharmaceutical Canada, Inc, located in Mirabel, QC, Canada. The product is available only through a prescription (Rx only) and distributed by Agnitio Inc. The lot number and expiration date are available but not provided in the text.*
atorvastatin-40mg500s - atorvastatin 40mg500s
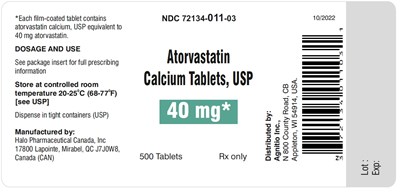
Each tablet contains 40mg of atorvastatin calcium, USP. The dosage and usage information can be found in the package insert. The tablets should be stored at a controlled temperature of 20-25°C (68-77°F) and dispensed in tight containers. The manufacturer is Halo Pharmaceutical Canada, Inc. located in Mirabel, Canada. The tablets are labeled with the NDC code 72134-011-03 and an expiration date of 10/2022.*
atorvastatin-40mg90s - atorvastatin 40mg90s
Atorvastatin Calcium Tablets, USP with 40mg atorvastatin equivalent is manufactured by Halo Pharmaceutical Canada, Inc. It comes in a pack of 90 film-coated tablets that should be stored at 20-25°C temperature. The prescribing information is provided in the package insert. Agnitio Inc is the distributor of this drug in the USA. The lot and expiry details are mentioned on the package.*
atorvastatin-80mg1000s - atorvastatin 80mg1000s

This is a prescription drug called Atorvastatin Calcium Tablets which are film-coated and contain 80 mg of atorvastatin calcium. It must be stored at room temperature and dispensed in tight containers. The package insert should be consulted for complete prescribing information. It comes in a bottle of 1000 tablets and is distributed by Agnitio Inc. with a manufacturing location in Canada. Lot and expiration date information are also provided.*
atorvastatin-80mg30s - atorvastatin 80mg30s

Atorvastatin Calcium Tablets, USP come in a film-coated form of 80mg per tablet. The package insert contains information about dosage and usage. The tablets should be stored at a temperature of 20-25°C and dispensed in tight containers. They are manufactured in Canada and distributed by Agnitio Inc. in the United States. The NDC number is 72134-012-01, and the expiry date is 10/2022. Lot number and manufacturing date are not available.*
atorvastatin-80mg500s - atorvastatin 80mg500s

This is a medication called Atorvastatin Calcium Tablets, USP containing 80 mg atorvastatin per film-coated tablet. It should be stored at a controlled room temperature of 20-25°C (68-77°F) and dispensed in tight containers. Further information and dosages can be found in the package insert. The medication is manufactured by Halo Pharmaceutical Canada, Inc and distributed by Agnitio Inc. This bottle contains 500 tablets and is only available with a prescription. The lot and expiration date are also indicated on the bottle.*
atorvastatin-80mg90s - atorvastatin 80mg90s
Atorvastatin Calcium Tablets, USP is a prescription medicine used for the treatment of high cholesterol levels in the body. Each tablet contains 80 mg of atorvastatin calcium, USP. It is recommended to store the tablets at room temperature between 20-25°C (68-77°F) and in tight containers. Complete dosage and usage should be taken as per the package insert. These tablets are manufactured by Halo Pharmaceutical Canada, Inc, and distributed by Agnitio Inc., USA. NDC code for this product is 72134-012-02. The lot number and expiry date are also mentioned on the package.*
atorvastatin-figu3 - atorvastatin figu3
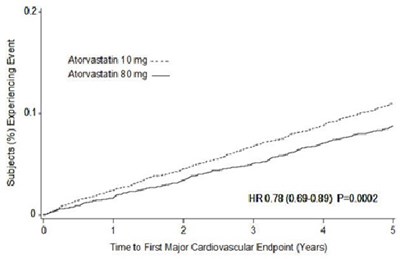
The text describes a graph or chart showing the percentage of subjects who experienced a major cardiovascular event after taking either 10mg or 80mg of Atorvastatin. It also provides a statistical analysis of the results.*
atorvastatin-figure2 - atorvastatin figure2
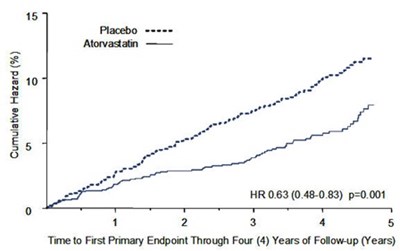
This is a graph depicting the cumulative hazard (%) for a medical study that lasted for four years. The hazard ratio (HR) for the study was 0.63 with a confidence interval of 0.48 - 0.83 and a p-value of 0.001. The x-axis displays the time to the first primary endpoint in years.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.