Product Images Alfuzosin Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Alfuzosin Hydrochloride NDC 72162-1449 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure-2 - figure 2

This text provides data comparing the efficacy of Alfuzosin hydrochloride extended-release tablets with a placebo in a clinical trial. The information includes the number of participants in each group (N=167 for placebo and N=170 for Alfuzosin), the timeline of treatment (0, 28, 56, 84 days), and a statistically significant result (***p< 0.001) for the mean change from baseline in IPSS Total Symptom Score during Trial 1.*
figure-3 - figure 3

Mean change in IPSS Total Symptom Score is compared between Placebo (N=152) and Alfuzosin hydrochloride extended-release tablets (N=137) after 28 and 56 days of treatment in Trial 2. The statistical significance is indicated as **p < 0.01.*
figure-4 - figure 4
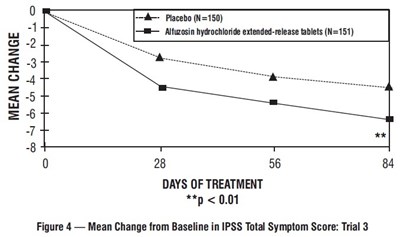
This document provides data on the mean change from the baseline in the International Prostate Symptom Score (IPSS) Total Symptom Score over a 28 to 56 days treatment period. The study compared the effects of a placebo group of 150 individuals with a group taking Alfuzosin hydrochloride extended-release tablets consisting of 151 individuals. The results show a statistically significant difference with a p-value of less than 0.01. This information is likely part of Trial 3 of a clinical trial or study.*
figure-5 - figure 5

This text provides information on mean change from baseline in peak urine flow rate (mL/s) over 56 days of treatment with placebo and alfuzosin hydrochloride extended-release tablets. The study included 167 patients in the placebo group and 170 patients in the alfuzosin group, showing a significant difference in peak urine flow rate.*
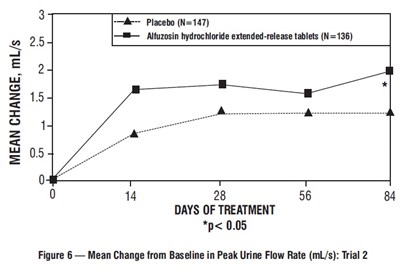
figure-7 - figure 7

This is a comparison of the mean change from baseline in peak urine flow rate for a trial involving Placebo (N=150) and Alfuzosin hydrochloride extended-release tablets (N=151) over 84 days of treatment. The data shows that there was no significant difference between the two groups (p > 0.05).*
Label - lbl721621449

This is a description of Alfuzosin Hydrochloride extended-release tablets containing 10 mg per tablet. The medication is stored in temperatures between 15° to 30°C. It is recommended to dispense in a tight, light-resistant container, protect from light and moisture, and keep out of reach of children. The usual dosage and other important information can be found in the package insert available at a specific website. The tablets should be swallowed whole and not chewed or crushed.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.