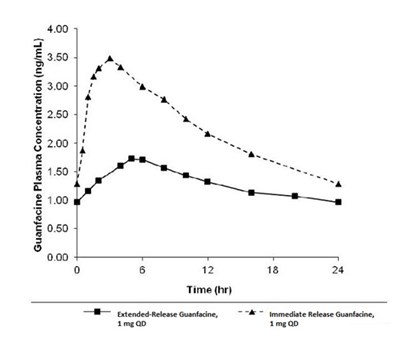
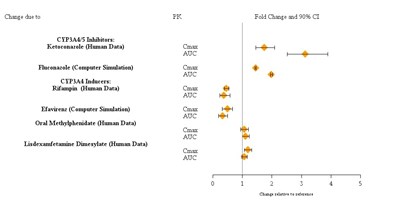
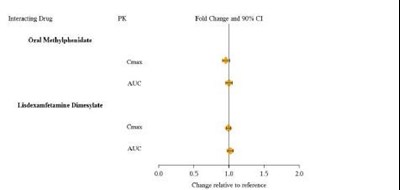
Product Images Guanfacine Extended-release
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Guanfacine Extended-release NDC 72162-2030 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label - lbl721622030

This is a medication description for tablets containing Guanfacine Hydrochloride. Each tablet comprises 4 mg of Guanfacine. The tablets should not be crushed, chewed, or broken before swallowing. The medication guide ought to be dispensed to each patient. The tablets are only available by prescription and should be stored between 15°C to 30°C in tightly sealed containers away from moisture. These tablets are manufactured by Apotex Inc and relabeled by Bryant Ranch Prepack, Inc. The product NDC number is 72162-2030-1. The FDA has information about this extended-release medication on their website.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.