Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 15 images provide visual information about the product associated with Donepezil Hydrochloride NDC 72162-2136 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. Time-course of the Change from Baseline in ADAS-cog Score for Patients Completing 24 Weeks of Treatment. - donepezil fig1
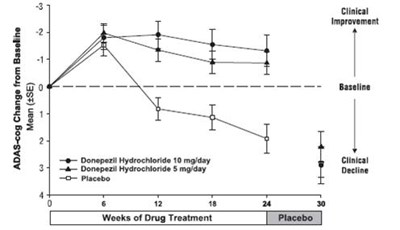
Figure 10. Cumulative Percentage of Patients Completing 6 Months of Double-blind Treatment with Particular Changes from Baseline in ADCS-ADL-Severe Scores. - donepezil fig10

The given text is not available.*
Figure 11. Time-course of the Change from Baseline in SIB Score for Patients Completing 24 Weeks of Treatment. - donepezil fig11

Figure 12. Cumulative Percentage of Patients Completing 24 Weeks of Double-blind Treatment with Specified Changes from Baseline SIB Scores. - donepezil fig12

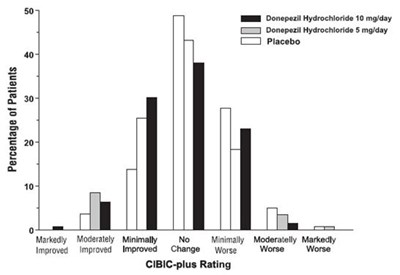
Figure 2. Cumulative Percentage of Patients Completing 24 Weeks of Double-blind Treatment with Specified Changes from Baseline ADAS-cog Scores. The Percentages of Randomized Patients who Completed the Study were: Placebo 80%, 5 mg/day 85%, and 10 mg/day 68%. - donepezil fig2

Figure 4. Time-course of the Change from Baseline in ADAS-cog Score for Patients Completing the 15-week Study. - donepezil fig4

Figure 5. Cumulative Percentage of Patients with Specified Changes from Baseline ADAS-cog Scores. The Percentages of Randomized Patients Within Each Treatment Group Who Completed the Study Were: Placebo 93%, 5 mg/day 90%, and 10 mg/day 82%. - donepezil fig5

Figure 7. Time Course of the Change from Baseline in SIB Score for Patients Completing 6 Months of Treatment. - donepezil fig7

Figure 8. Cumulative Percentage of Patients Completing 6 Months of Double-blind Treatment with Particular Changes from Baseline in SIB Scores. - donepezil fig8

Figure 9. Time Course of the Change from Baseline in ADCS-ADL-Severe Score for Patients Completing 6 Months of Treatment. - donepezil fig9

Label - lbl721622136

This appears to be the description of a medication called Donepezil Hydrochloride. Each tablet contains 5 mg of Donepezil. The National Drug Code (NDC) for this medication is 72162-2136-3. It is recommended to store the medication at a temperature between 20°C to 25°C (68°F to 77°F), with excursions permitted between 15°C to 30°C (59°F to 86°F). The medication should be dispensed in a tight, light-resistant container with a child-resistant closure. It is important to keep this and all medications out of the reach of children. The medication should be accompanied by a medication guide for each patient.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.