Product Images Tamsulosin Hydrochloride
View Photos of Packaging, Labels & Appearance
- Label - lbl721622509
- Figure 1 Mean Plasma Tamsulosin Hydrochloride Concentrations Following Single-Dose Administration of Tamsulosin Hydrochloride Capsules 0.4 mg Under Fasted and Fed Conditions (n=8) - tamsulosin fig1
- Figure 2A Mean Change from Baseline in Total AUA Symptom Score (0 to 35) Study 1 - tamsulosin fig2
- Figure 2B Mean Change from Baseline in Total AUA Symptom Score (0 to 35) Study 2 - tamsulosin fig3
- Figure 3A Mean Increase in Peak Urine Flow Rate (mL/Sec) Study 1 - tamsulosin fig4
- Figure 3B Mean Increase in Peak Urine Flow Rate (mL/Sec) Study 2 - tamsulosin fig5
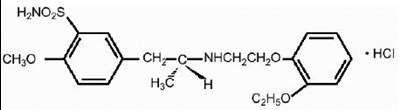
- Chemical Structure - tamsulosin str
Product Label Images
The following 7 images provide visual information about the product associated with Tamsulosin Hydrochloride NDC 72162-2509 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label - lbl721622509

Each capsule contains Tamsulosin Hydrochloride USP 0.4 mg. The usual dosage instructions and prescribing information can be found in the package insert. Store the capsules at a temperature of 20° to 25°C (68° to 77°F), avoiding excessive moisture. They must be dispensed in a tight container and kept out of reach of children. This product, containing 100 capsules, is relabeled by Bryant Ranch Prepack, Inc. and manufactured by Aurobindo Pharma Limited.*
Figure 1 Mean Plasma Tamsulosin Hydrochloride Concentrations Following Single-Dose Administration of Tamsulosin Hydrochloride Capsules 0.4 mg Under Fasted and Fed Conditions (n=8) - tamsulosin fig1

This text provides information on the mean plasma concentration of Tamsulosin Hydrochloride following the administration of 0.4 mg capsules under both fasted and fed conditions. The concentration data is likely presented graphically in Figure 1 over a period of time in hours.*
Figure 2A Mean Change from Baseline in Total AUA Symptom Score (0 to 35) Study 1 - tamsulosin fig2

Figure 2A shows the mean change from baseline in Total AUA Symptom Score (on a scale of 0 to 35) for Study 1. The figure compares the treatment groups with tamsulosin 0.08mg, alfuzosin 10mg, and placebo over a duration of weeks. The data includes the number of participants for each treatment group at different time points.*
Figure 2B Mean Change from Baseline in Total AUA Symptom Score (0 to 35) Study 2 - tamsulosin fig3
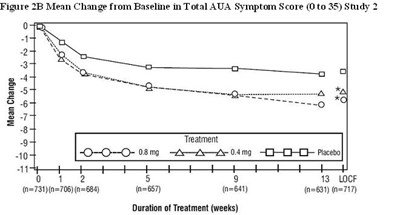
This is a statistical data related to the treatment with different doses of LNt (0.08mg to 0.4mg) and placebo in Study 2, showing the mean change from baseline in the Total AUA Symptom Score over a duration of treatment in weeks.*
Figure 3A Mean Increase in Peak Urine Flow Rate (mL/Sec) Study 1 - tamsulosin fig4
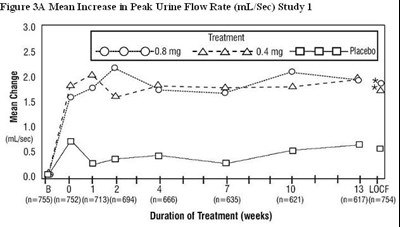
This is a study evaluating the mean increase in peak urine flow rate for different treatments over a specified duration in weeks. The results are presented in Figure 3A and include the treatments of 25mg, 0.08mg, 2x0.4mg, and Placebo. The study involved a significant number of participants across different treatment groups.*
Figure 3B Mean Increase in Peak Urine Flow Rate (mL/Sec) Study 2 - tamsulosin fig5
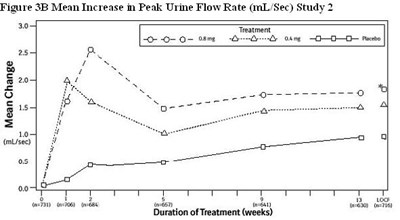
The text provides information about the Mean Increase in Peak Urine Flow Rate (mL/Sec) in Study 2, specifically dealing with different treatment options and the Duration of Treatment in weeks. The data seems to be related to a scientific study on urinary flow rates.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.