Product Images Orserdu
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Orserdu NDC 72187-0102 by Stemline Therapeutics, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - elacestrant 02
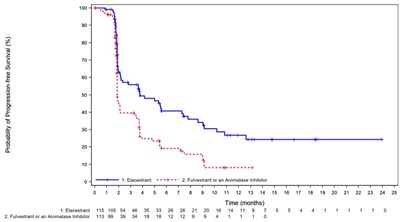
This is a graph showing the probability of progression-free survival (%) over time (months) for two treatments: T-Ecestrant and Fulvostrant, an Aromatase Inhibitor. The graph shows the time across the X-axis and the probability of progression-free survival across the Y-axis.*
Orserdu Bottle Label - 86mg - elacestrant 03
This is a description of a medication called elacestrant, which comes in tablet form with a dosage of 86 mg. The tablets are manufactured by Stemline Therapeutics and are for prescription use only. The packaging includes the company's contact information, manufacturing date, and recommended storage conditions. The medication should be kept out of reach of children.*
Orserdu Bottle Label - 345mg - elacestrant 04

This is a description of a medication named "Elacestrant" manufactured by Stemline Therapeutics, Inc. The medication comes in tablet form, with each tablet containing 345 mg of Elacestrant, which is equivalent to 400 mg of Elacestrant dihydrochloride. It is recommended for use only as per the prescription. The medication should be stored at 20°C to 25°C (68°F to 77°F) with permitted excursions to 15°C to 30°C (59°F to 86°F). The NDC code for this medication is 72187-0102-3, and it is recommended to keep it out of reach of children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
