Product Images Xpovio
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 38 images provide visual information about the product associated with Xpovio NDC 72237-104 by Karyopharm Therapeutics Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
kar00 0014 04

This is a description of weekly dosage instructions for XPOVIO (selinexor) tablets. The blister pack contains three tablets of 20mg each. To open, the user must press and hold a button, then pull out the medication card. The tablets should not be broken, chewed, crushed, or divided. Each patient should also be given a medication guide. The rest of the text is not legible.*
kar00 0014 05

The text describes a medication called XPOVIO tablets (selinexor) manufactured for Karyopharm Therapeutics. The tablets are to be pushed through foil and swallowed whole with water. The medication should only be taken as prescribed by a physician, with only one 60 mg dose per week. Dosing instructions can be found in the prescribing information. Lot and expiration numbers are included but are not readable in the provided text.*
kar00 0014 06
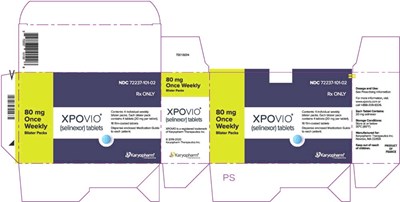
Description: Selinexor is a medication available in tablet form with a strength of 80mg. It is administered once a week and is available under the brand name XPOVIO. The NDC number of this medication is 7223710102. Any other information regarding this medication is not available from the given text.*
kar00 0014 07

This is a description of a prescription drug called "XPOVIO" that comes in a blister pack containing four tablets per pack, each tablet containing 20mg. The medication guide enclosed in the pack should be dispensed to every patient. The dosing information should be reviewed in the prescribing information. The instruction for opening the blister pack is included. There is some illegible or unclear characters, but the drug appears to be taken once weekly at 80mg.*
kar00 0014 08

This is the packaging information for a medication called XPOVI o (selinexor) tablets, manufactured for Karyopharm Therapeutics Inc. There are instructions for taking the medication, including pushing the tablets through the foil and swallowing them whole with water. It is important to take the medication as prescribed by a physician, including taking only one 80 mg dose in a single day per week. Dosing instructions can be found in the prescribing information. Lot and expiration information is shown but are obscured for privacy reasons.*
kar00 0014 09

This is a prescription for XPOVIO medication. The recommended dosage is 60 mg taken twice a week. The medication comes in the form of tablets with the NDC 7223710103. The prescription also mentions some other medications, including BIGRCEIN and selinexon.*
kar00 0014 10

This is a prescription drug blister pack containing 60 tablets of 20 mg doses (totaling 120 mg weekly), with instructions to be administered twice weekly. It is important to refer to the enclosed Prescribing Information for proper dosing instructions. The medication is called XPOVIO and care should be taken to not break, chew, or crush the tablets. The enclosed medication guide should be dispensed to each patient. The listed text following the instructions appears to be illegible and cannot be used for further description.*
kar00 0014 11

This is a description of a medication called XPO\/lO (selinexor) tablets manufactured for Karyopharm Therapeutics Inc. The tablets need to be pushed through the foil before swallowing with water. It's important to take each dose as prescribed by a physician and only take a maximum of one 60 mg dose per day. The recommended dosing instructions can be found in the Prescribing Information.*
kar00 0014 13

This is a prescription medication contained in a blister pack. The pack contains 80 tablets each of 20 mg. It is advised to refer to prescribing information for dosing instructions. Breaking, chewing or crushing the tablets is not recommended. The text also includes some alphanumeric codes which may relate to the medication, instructions or other aspects of the packaging.*
kar00 0014 14

This is a description of a medication called XPOVIO (selinexor) tablets. It is manufactured for Karyopharm Therapeutics Inc. in Newton, MA, with a lot number of 000000 and expiration date of XXX0000. The tablets should be pushed through foil and taken whole with water, following the prescribed dosage of one 80 mg dose on Day 1 and Day 3 of the week. It is important to refer to the Prescribing Information for specific dosing instructions and to only take one 80 mg dose in a single day.*
kar00 0014 15

XPOVIO (selinexor) tablets are available in a blister pack and come in a strength of 100mg. They are to be taken once a week. The National Drug Code for this medication is 7223710105. This medication is only available through a prescription.*
kar00 0014 16

XPOVIO (selinexor) tablets for prescription use are packed in a blister pack of 5 tablets (20 mg each). The enclosed medication guide should be distributed to each patient. The instructions on how to open the package should be followed carefully. Please refer to the Prescribing Information for dosing instructions. Do not chew, break, crush, or divide the tablets. The other text is not clearly readable.*
kar00 0014 17

This text is a set of instructions for taking XPOVIO tablets manufactured for Karyopharm Therapeutics Inc. It provides information about the dosage, usage, and administration of the medicine. The text also includes the LOT and EXP numbers.*
kar00 0014 18

This is a description of two National Drug Codes (NDC) which are 7223740106 and 7223710106. The prescription is for RXONLY medication. The name of the medication is XPOVIO which is a weekly oral tablet with the generic name of selinexor. The medication is produced by T HE Rl and is available in two strengths: fomooyubes and Seimzinmion. The dosage of the tablets is not given.*
kar00 0014 19

This is a medication prescription container for Trm e XPOVIO with a weekly dose of 40mg. The container includes a blister pack that has four tablets, each containing 20mg of Selinexor. The instructions on how to open the container are given in two steps with accompanying images. The container also has a warning that the tablets should not be broken, chewed, crushed, or divided. An enclosed medication guide is to be dispensed to each patient. The rest of the document consists of non-English characters and is therefore not useful.*
kar00 0014 20

This appears to be a label for a medication called XPOVIO (selinexon tablets) manufactured for Karyopharm Therapeutics Inc. The medication comes in 40mg doses and is to be taken as prescribed by a physician. The label includes instructions to push tablets through the foil and swallow whole with water. The lot number is 000000 and the expiration date is XXX0000.*
kar00 0014 22

This is a blister pack of 40mg tablets administered weekly. The package contains 2 tablets with 20mg each. There are instructions on how to open the pack properly, and there is a note to refer to the prescribing information for dosing. No other valuable information is available.*
kar00 0014 23

XPOVIO is a medication available in tablet form, specifically selinexon tablets. The recommended dose is one 40mg tablet per week, as prescribed by a physician. The tablets should be pushed through the foil and swallowed whole with water. Additional dosing instructions can be found in the prescribing information.*
kar00 0014 24

XPOVIO is a prescription medicine used to treat certain types of lymphoma. It is available in the form of tablets with a strength of 40mg. The medicine is taken weekly and is not available without a prescription. The text also includes some reference codes and trademarks.*
kar00 0014 25

This is a medication blister pack labeled NDC 72237-102-17. Each blister contains one tablet with 40mg of selinexor, marked with the inscription "XPOVIO". Patients should refer to the prescribing information for dosage instructions. The medication should be dispensed following the enclosed guide to each patient. The pack includes instructions with images detailing how to correctly open it.*
kar00 0014 26

This is a description for XPOVIO selinexor tablets by Karyopharm Therapeutics Inc. The package contains one 40 mg dose and has a LOT number and EXP date. The user should take only one 40 mg dose per week as prescribed by a physician. The tablets should be pushed through the foil and swallowed whole with water. For more information on dosing instructions, refer to the Prescribing Information.*
kar00 0014 28

This is a medicine package for a drug called "Xpovio" by Karyopharm Therapeutics. The medication comes in the form of 16 tablets, where the recommended dosage is one tablet weekly. The blister pack has instructions printed on it, which include pressing a button to remove the medication card. The package contains a medication guide that should be given to each patient. The rest of the text is not entirely readable.*
kar00 0014 29

This is a medication package of XPOVIO selinexon tablets, manufactured for Karyopharm Therapeutics Inc. in Newton, MA, USA. The lot number is 00000000, and the expiration date is XXX0000. Each tablet contains 40mg of the active ingredient, and the dosing instructions are to take one 40mg tablet on Day 1 and one on Day 3 of the week, as prescribed by a physician. The tablets should be swallowed whole with water after pushing them out of the foil.*
kar00 0014 30

This is a prescription medication called XPOVIO, which is available in a dosage of 60 mg. It is taken once a week, and comes in the form of enteric-coated tablets. The NDC (National Drug Code) for this medication is 7223710401, which is listed twice on the packaging. The text also includes the names "RXONLY" and "&Karyopharm", but these do not provide additional information about the medication itself.*
kar00 0014 31

This is a description of a medication called XPOVIO, whose generic name is selinexor. The medication comes in the form of tablets, and one blister pack contains one 60mg tablet. The pack has instructions on how to open it, which involves pressing a button and pulling out the medication card. This medication guide should be given to each patient. The text also warns that the tablets should not be broken, chewed, crushed or divided. However, full dosing instructions are included in the prescribing information. The text also includes a reference to the manufacturer, Karyopharm Therapeutics, and the product is only available with a prescription.*
kar00 0014 32

This is a product label for XPOVIO selinexon tablets manufactured by Karyopharm Therapeutics Inc. The label indicates important instructions for use, including taking only one 60 mg dose per week as prescribed by a physician, pushing the tablet through its foil packaging, and swallowing the tablet whole with water. The label also includes information on lot number and expiration date.*
kar00 0014 35

This text is a set of instructions for taking XPOVIO (selinexor) tablets, including some manufacturing and contact information. The instructions also specify the dosage and frequency of the medication. The tablet is meant to be taken once a week, and each dose is one 80 mg tablet. The patient should take the entire pack and should follow the dosing instructions in the Prescribing Information. The tablet should be swallowed whole with water, and the patient should push the tablet through the foil before taking it.*
kar00 0014 37

This is a blister pack for 100 mg XPO salinexor tablets. It contains 2 tablets (50 mg per tablet) and comes with a medication guide for each patient. The pack can be opened by pressing and pulling the medication card. The prescribing information for dosing should be referred to. The rest of the text is not available.*
kar00 0014 38

XPOVI O® (selinexor) tablets are manufactured for Karyopharm Therapeutics Inc. The packaging includes the LOT and EXP information indicating the batch and expiration date respectively. The tablets are intended for oral administration with a recommended 100 mg dose per week as per the prescribing information. The tablets should be taken whole with water after pushing it through the foil.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.