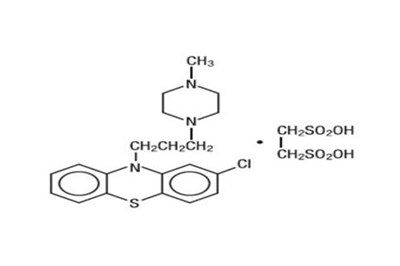
Product Images Prochlorperazine Edisylate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Prochlorperazine Edisylate NDC 72266-204 by Fosun Pharma Usa Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Prochlorperazine Carton Label - Prochlorperazine Carton Label

The text describes the details of Prochlorperazine Edisylate Injection, USP, a drug used for deep intramuscular or intravenous use only, not for subcutaneous use. It comes in 10x 2mL Multiple-Dose Vials. Each vial contains Prochlorperazine, USP, 5mg/mL, Monobasic Sodium Phosphate Monohydrate, Sodium Tartate Dihydrate, Saushan Sodium, Byl Aotl, and Water for Ijection. The drug is not to be used in pediatric surgery. Fosun Pharma USA distributes the drug, which is made in India. MaiDiscolored ismarkedly, contents should be discarded.*
Prochlorperazing Edisylate Injection Vial Label - Prochlorperazine Label

Prochlorperazine Edisylate Injection is distributed by Fosun Pharma USA Inc. in Princeton, NJ. This medication is made in India and is available in a 2 mL multiple-dose vial that is only for deep IM or IV use, not for SC use. The vial contains 10 mg/2 mL (5 mg/mL) of the medication. The package bears the warning to protect it from light. The rest of the text is unavailable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.