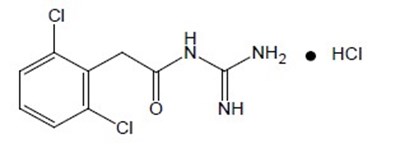
Product Images Guanfacine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Guanfacine NDC 72266-258 by Fosun Pharma Usa, Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - guanfacine 02

This text provides information about two medications: Tenex and Guanfacine extended-release tablets, both at a dosage of 1 mg once a day. The data also includes numbers such as 3.50, 0, 5, 24, 18, and 12 which may refer to different parameters or measurements. Time in hours is shown on the graph with values ranging from -4 to 12. *
Figure 2 - guanfacine 03

This description contains information about drugs affecting CYP3A4 enzymes, including inhibitors like ketoconazole and fluconazole, as well as inducers like rifampin and efavirenz. It also mentions oral methylphenidate and lisdexamfetamine dimesylate with Cmax and AUC data. The text refers to changes compared to a reference, likely in terms of PK Fold Change and 90% CT.*
Figure 3 - guanfacine 04

This text appears to contain information related to the pharmacokinetics of two drugs: Oral Methylphenidate and Lisdexamfetamine Dimesylate. It includes terms such as "Cmax" (maximum plasma concentration) and "AUC" (area under the curve), as well as references to fold change and 90% confidence intervals. The text seems to provide data on the interaction and effects of these drugs.*
1mg - guanfacine 06

This is a description of Guanfacine Extended-Release Tablets, USP. The tablets should not be crushed, chewed, or broken before swallowing. Each tablet contains guanfacine. It is recommended to store the tablets at a temperature between 20°C to 25°C, with excursions permitted up to 30°C. The product is distributed by Fosun Pharma USA Inc., with manufacturing in China. Please refer to the package insert for complete prescribing information.*
2 mg - guanfacine 07

This text contains information related to pharmaceuticals, specifically mentioning a package insert for complete prescribing information. It includes details about tablets, their storage conditions, and precautions about how the tablets should be taken. The product seems to be extended-release tablets by Fosun Pharma.*
3 mg - guanfacine 08

This text provides information about guanfacine extended-release tablets that should not be crushed, chewed, or broken before swallowing. It also mentions the tablets containing FD&C Yellow No. 6 as a color additive and advises storing them at a specific temperature range. The text indicates that each tablet contains 3 mg of guanfacine and includes information on the distributor and batch details. Additionally, it recommends referring to the package insert for complete prescribing information.*
4 mg - guanfacine 09

This text contains information about Guanfacine extended-release tablets, USP. Each tablet contains 4g of Guanfacine. It provides storage instructions, dosage recommendations, and details about the manufacturer and distributor. The tablets should not be crushed, chewed, or broken before swallowing. The tablets contain FD&C Yellow No. 5 as a color additive. The packaging includes 100 tablets.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.