Product Images Pemetrexed
View Photos of Packaging, Labels & Appearance
- PACKAGE CARTON – Pemetrexed for Injection 100 mg single-dose vial - pemetrexed 100mg carton trade
- PACKAGE CARTON – Pemetrexed for Injection 100 mg single-dose vial - pemetrexed 100mg label
- PACKAGE CARTON – Pemetrexed for Injection 500 mg single-dose vial - pemetrexed 500mg carton trade
- PACKAGE Label – Pemetrexed for Injection 500 mg single-dose vial - pemetrexed 500mg label
- Structural Formula - pemetrexed f01 v01
- Figure 1 - pemetrexed f01
- Figure 2 - pemetrexed f05 v04
- Figure 3 - pemetrexed f08 v3
- Figure5 - pemetrexed f12 v3
- Figure 6 - pemetrexed f13 v2
- Figure 8 - pemetrexed f14 v2
- Figure 4 - pemetrexed f15 v01
- Figure 7 - pemetrexed f16 v1
- Figure 8 - pemetrexed f17 v1
Product Label Images
The following 14 images provide visual information about the product associated with Pemetrexed NDC 72338-100 by Waverley Pharma Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE CARTON – Pemetrexed for Injection 100 mg single-dose vial - pemetrexed 100mg carton trade

Pemetrexed For Injection is an intravenous cytotoxic agent used with usual dosage instructions detailed in accompanying literature. The vial contains pemetrexed disodium and mannitol, with hydrochloric acid and/or sodium hydroxide to balance the pH. The powder is stored at room temperature, and the reconstituted solution must be further diluted before use, and refrigerated. The product is available in single-dose packaging and must be discarded if any unused portion is left. The drug is serialized, and the provider of the drug is Medicure Pharma, Inc. The drug is manufactured in India.*
PACKAGE CARTON – Pemetrexed for Injection 100 mg single-dose vial - pemetrexed 100mg label

This is a description of a cytotoxic agent called Pemetrexed Disodium. Each vial has a capacity of 100mg of Pemetrexed and 106mg of mannitol. The pH may have been adjusted using hydrochloric acid and/or sodium hydroxide. The powder should be stored at 25°C with a permitted excursion to 15-30°C. The medication is for intravenous use only, and the accompanying literature should be consulted for reconstituted and infusion solution storage. The details of the distributor, Medicure Pharma Inc., are also included.*
PACKAGE CARTON – Pemetrexed for Injection 500 mg single-dose vial - pemetrexed 500mg carton trade

This is a drug information sheet for Pemetrexed For Injection, USP. It provides instructions on how to reconstitute and use the drug, the usual dosage, and storage requirements. The drug is a cytotoxic agent and caution should be taken when handling it. The product is manufactured in India and distributed in the US by Medicure Pharma, Inc. The sheet also contains product and classification codes, such as NDC code 72338-101-01, and a serialization code.*
PACKAGE Label – Pemetrexed for Injection 500 mg single-dose vial - pemetrexed 500mg label

This is a medication with NDC 72338-101-01 that comes in a vial containing pemetrexed disodium equivalent to 500mg pemetrexed, 500mg of mannitol, and may have hydrochloric acid and/or sodium hydroxide added to it. It is a cytotoxic agent to be stored at 25°C, with excursions permitted to 15-30°C, for intravenous use only. It is distributed by Medicure Pharma, Inc. and made in India. No information is given about its usage or indications.*
Figure 1 - pemetrexed f01

This appears to be a graph that shows the overall survival rate over time for a treatment arm using AsPiC and Asc. The x-axis measures time in months and the y-axis measures survival percentage. At each time point, the number of patients at risk is also shown.*
Figure 2 - pemetrexed f05 v04

The text shows a graph plotting the survival of patients on chemotherapy drugs Pemetrexed + Cisplatin (AC) and Gemcitabine + Cisplatin (GC) over a period of 30 months. The x-axis measures survival time in months, and the y-axis is the probability of survival. The number of patients at risk is also included for each drug. The graph shows that at 30 months, the survival probability for AC and GC was 0% as all patients had died.*
Figure 3 - pemetrexed f08 v3

This appears to be a survival probability chart showing the survival time of patients who were administered Pemetrexed + Cisplatin and Gemcitabine + Cisplatin. The chart also includes patient counts denoted as "patients at risk". There is no additional information available.*
Figure 6 - pemetrexed f13 v2

Not available. The text seems to be truncated and there is not enough information to generate a useful description.*
Figure 8 - pemetrexed f14 v2
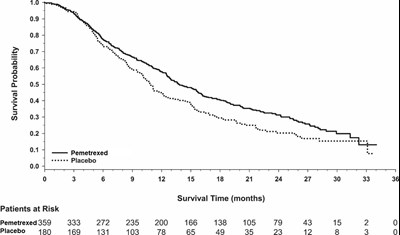
This is a graph depicting the survival probability of patients who received Pemetrexed and those who received Placebo over a period of 36 months with survival time in months marked on the X-axis and survival probability on the Y-axis. The graph shows the number of patients who were still alive or had died at various time points. The number of patients at risk for each group is also provided.*
Figure 7 - pemetrexed f16 v1

This text appears to be a table with two columns titled "Pemetrexed" and "Placebo". The first row seems to be a header for "Survival Probability" followed by empty spaces. In the second row, there are the headers "Survival Time (months)" and "Patients at Risk". The remaining rows seem to contain numeric values, possibly indicating the survival time in months for patients receiving Pemetrexed and Placebo treatments, as well as the number of patients being studied. However, due to the poor quality of the text and missing characters, a complete evaluation of the provided text cannot be made.*
Figure 8 - pemetrexed f17 v1

This is a survival probability table showing the number of patients at risk for Pemetrexed + Cisplatin and Cisplatin treatments over time in months. The data is presented in a chart.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


