Product Images Memantine Hydrochloride
View Photos of Packaging, Labels & Appearance
- Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 01
- Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 02
- Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 03
- Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 04
- Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 05
- Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 06
- Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 07
- Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 08
- Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 09
- Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 10
- Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 11
Product Label Images
The following 11 images provide visual information about the product associated with Memantine Hydrochloride NDC 72578-004 by Viona Pharmaceuticals Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 07
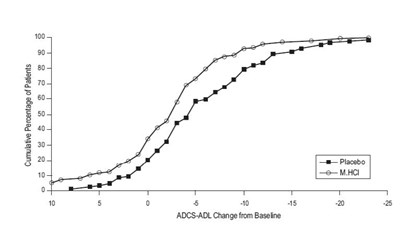
The text provides a graph showing the cumulative percentage of patients related to two different drugs: Placobol/Donopozil and M.HClDonepezil. It also shows a line graph representing the change from Baseline score for the Alzheimer's Disease Cooperative Study-Activities of Daily Living Scale (ADCS-ADL) with a value of 25.*
Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 09
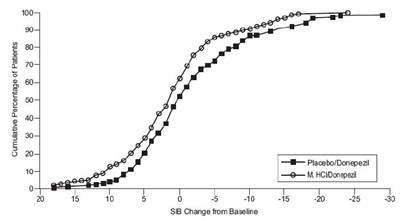
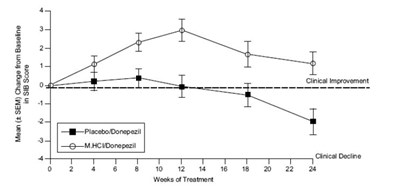
This is a graph that shows the cumulative percentage of patients and their SIB (Severe Impairment Battery) change from baseline while taking either a placebo or Donepezil (M HCI) medication. The graph shows that the patients taking Donepezil had a higher percentage of positive SIB changes compared to those on the placebo.*
Memantine Hydrochloride Tablets - 5b56b880 7a88 4d48 8291 96f38c791808 11
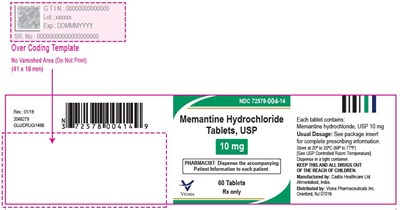
Memantine Hydrochloride is a medication available in tablet form with each tablet containing 10mg of the compound. The medication is Rx-only and should be dispensed along with Patient Information by the attending pharmacist according to guidelines provided in the package insert. The tablets must be stored in a controlled room temperature and kept out of children's reach. They are packaged in a container size of 60 tablets. The manufacturer is Cadia Healthcare Limited, located in India, and the distributor is Vona Pharmaceuticals, based in Maryland, USA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.