Product Images Bortezomib
View Photos of Packaging, Labels & Appearance
Product Label Images
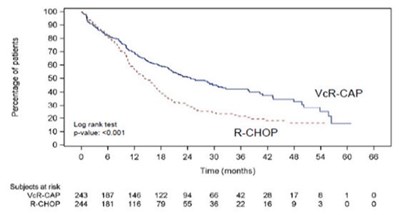
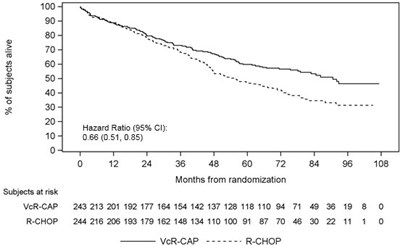
The following 9 images provide visual information about the product associated with Bortezomib NDC 72603-270 by Northstar Rxllc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
cartonlabel - cartonlabel

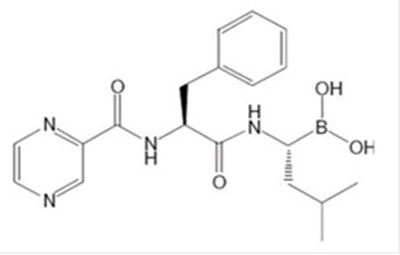
This text provides important information about a pharmaceutical product, including details such as the expiration date, lot number, serial number, and 2D barcode containing GTIN. The product is a lyophilized vial containing bortezomib and manufactured by Dr. Reddy's Laboratories Limited. Instructions for reconstitution and administration of the medication are given. Additionally, storage and dosage information is included, emphasizing the need for proper handling and dilution of the product, which is a cytotoxic agent.*
figure3 - figure3
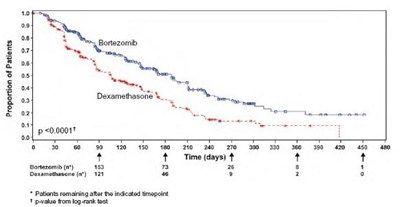
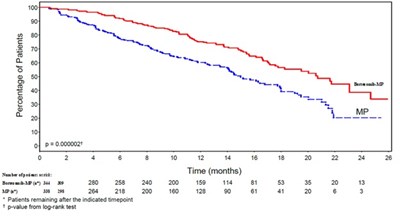
The text appears to contain information related to the proportion of patients in a study, with a significant P-value of less than 0.0001. It seems to also mention something about the number of patients remaining or indicated at a specific time point of 4 days. It may be related to a medical or clinical study, but additional context is needed for a more specific evaluation.*
figure4 - figure4
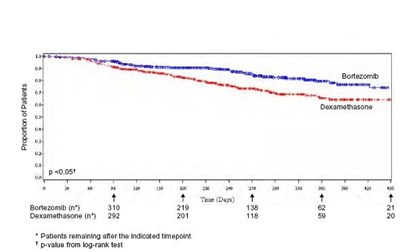
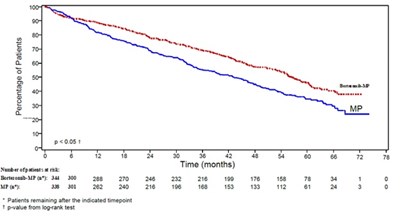
This text provides information about a report on patients treated with Bortezomib and Dexamethasone. It includes data on the number of patients remaining at specific timepoints and mentions the p-value from a log-rank test. The comparison between the two treatments can be evaluated using this data.*
viallabel - viallabel

This text provides essential information on a medication called Bortezomib, which is presented in a lyophilized vial containing 3.5 mg of the active ingredient along with 35 mg of mannitol. The suggested dosage details can be found in the accompanying package insert. The storage conditions specify a temperature range of 20°C to 25°C and emphasize protection from light. The product is intended for intravenous use without any preservatives added. The vial is identified by a specific code, and any unused portion should be discarded following the guidelines provided. The medication is manufactured by Dr. Reddy's Laboratories Limited in Visakhapatnam, India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.