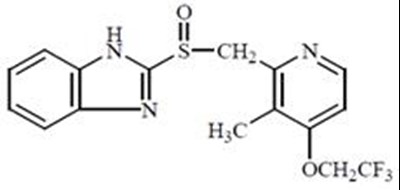
Product Images Lansoprazole
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Lansoprazole NDC 72603-314 by Northstar Rx Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure1 - lansoprazole fig1

This is a description of a study evaluating the mean severity of day heartburn for evaluable patients over time. The severity is ranked on a scale of 3 (severe) to 0 (none). The study compares the effects of Placebo, Lansoprazole 15 mg QD, and Lansoprazole 30 mg QD on day heartburn severity. The chart shows the progression of heartburn severity from mild to severe over days from the start of treatment.*
figure2 - lansoprazole fig2

This is a visual representation of the mean severity of night heartburn by study day for evaluable patients, comparing the effects of placebo, Lansoprazole 16mg QD, and Lansoprazole 30mg QD. The severity levels are categorized as 3=Severe, 2=Moderate, 1=Mild, and 0=None. The graph shows the progression of night heartburn severity over the course of treatment days.*
FIGURE - lansoprazole fig3
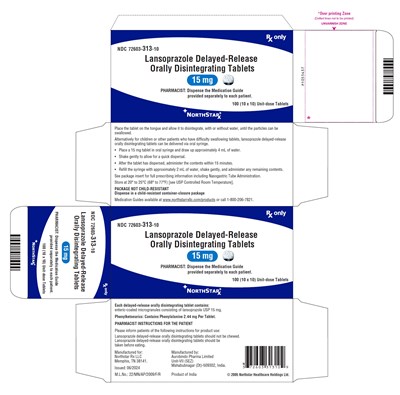
This is a detailed description of Lansoprazole Delayed-Release Orally Disintegrating Tablets provided by Northstar Rx LLC. These tablets are indicated for patients who have difficulty swallowing traditional tablets. The tablets should be placed on the tongue and allowed to disintegrate before swallowing, either with or without water. For children or patients who struggle with swallowing, an oral syringe can be used to administer the tablets by first dispersing it in water. It is important not to chew the tablets and to take them before eating. The tablets contain enteric-coated microgranules of lansoprazole USP 15 mg. This packaging includes 100 unit-dose tablets (10 x 10) and a Medication Guide for each patient. Instructions provided include storing the tablets at room temperature, and the manufacturer details are Northstar Rx LLC and Aurobindo Pharma Limited. Please visit www.northstarrxllc.com for more information.*
figure - lansoprazole fig4

This text provides detailed information about Lansoprazole Delayed-Release Orally Disintegrating Tablets. It includes instructions for administering the medication to patients, such as the recommended method of consumption, storage conditions, and a reminder that the tablets should not be chewed. The text also mentions the presence of Phenylalanine in the tablets and the manufacturer's details, including contact information and the origin of the product. Additionally, it outlines a process for the administration of the tablets for patients who have difficulty swallowing.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.