Product Images Heparin Sodium
View Photos of Packaging, Labels & Appearance
- Principal Display Panel – Heparin Sodium Injection, USP 1,000 USP units per mL Carton - 1000 1mL carton
- Principal Display Panel – Heparin Sodium Injection, USP 1,000 USP units per mL Vial Label - 1000 1mL container
- Principal Display Panel – Heparin Sodium Injection, USP 10,000 USP units per 10 mL Carton - 10000 10mL carton
- Principal Display Panel – Heparin Sodium Injection, USP 10,000 USP units per 10 mL Vial Label - 10000 10mL container
- Principal Display Panel – Heparin Sodium Injection, USP 10,000 USP units per mL Carton - 10000 1mL carton
- Principal Display Panel – Heparin Sodium Injection, USP 10,000 USP units per mL Vial Label - 10000 1mL container
- Principal Display Panel – Heparin Sodium Injection, USP 30,000 USP units per 30 mL Carton - 30000 30mL carton
- Principal Display Panel – Heparin Sodium Injection, USP 30,000 USP units per 30 mL Vial Label - 30000 30mL container
- Principal Display Panel – Heparin Sodium Injection, USP 5,000 USP units per mL Carton - 5000 1mL carton
- Principal Display Panel – Heparin Sodium Injection, USP 5,000 USP units per mL Vial Label - 5000 1mL container
- Principal Display Panel – Heparin Sodium Injection, USP 50,000 USP units per 10 mL Carton - 50000 10mL carton
- Principal Display Panel – Heparin Sodium Injection, USP 50,000 USP units per 10 mL Vial Label - 50000 10mL container
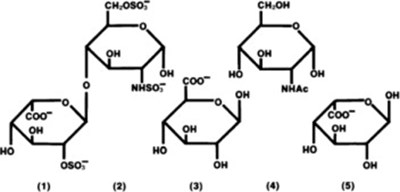
- Structure of Heparin Sodium - hep structure
Product Label Images
The following 13 images provide visual information about the product associated with Heparin Sodium NDC 72603-412 by Northstar Rxllc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Principal Display Panel – Heparin Sodium Injection, USP 1,000 USP units per mL Carton - 1000 1mL carton

Principal Display Panel – Heparin Sodium Injection, USP 1,000 USP units per mL Vial Label - 1000 1mL container

This is a description of a pharmaceutical product labeled NDC72603-234-01, identified for prescription use. The product contains heparin from porcine intestinal mucosa and is made in China for Northstar Rx LLC. It is a Sodium Injection, USP, with a concentration of 1,000 USP units per mL. The product is packaged in a 1 mL Single-Dose Vial for intravenous or subcutaneous use, with an expiration date of 02/2024.*
Principal Display Panel – Heparin Sodium Injection, USP 10,000 USP units per 10 mL Carton - 10000 10mL carton

Principal Display Panel – Heparin Sodium Injection, USP 10,000 USP units per 10 mL Vial Label - 10000 10mL container

Principal Display Panel – Heparin Sodium Injection, USP 10,000 USP units per mL Carton - 10000 1mL carton

NDC 7260350125 is a code for a pharmaceutical product that contains 100 units of USP (United States Pharmacopeia) of an unidentified substance. The text also includes some additional details in an unclear format, possibly related to dosage and administration instructions. Further evaluation may be needed for a complete understanding.*
Principal Display Panel – Heparin Sodium Injection, USP 10,000 USP units per mL Vial Label - 10000 1mL container

This is a product label for Heparin Sodium Injection, a medication used for intravenous or subcutaneous administration. It contains 10,000 USP units per mL and is not suitable for lock flush use. The medication is manufactured in China for Northstar Rx LLC and comes in a 1 mL single-dose vial. The expiration date is noted on the label as "Exp.: [date]".*
Principal Display Panel – Heparin Sodium Injection, USP 30,000 USP units per 30 mL Carton - 30000 30mL carton

The text is not-readable.*
Principal Display Panel – Heparin Sodium Injection, USP 30,000 USP units per 30 mL Vial Label - 30000 30mL container

Principal Display Panel – Heparin Sodium Injection, USP 5,000 USP units per mL Carton - 5000 1mL carton

Principal Display Panel – Heparin Sodium Injection, USP 5,000 USP units per mL Vial Label - 5000 1mL container

This text contains information about a medical product with the NDC code 72603-412-01, indicating it as a prescription-only medication made from Porcine Intestinal Mucosa in China. The product is Heparin Sodium Injection, USP, manufactured by Northstar Healthcare. This single-dose vial is intended for intravenous or subcutaneous use and should not be used for lock flush purposes. The expiration date is February 2024 with 500 USP units per mL.*
Principal Display Panel – Heparin Sodium Injection, USP 50,000 USP units per 10 mL Carton - 50000 10mL carton

Principal Display Panel – Heparin Sodium Injection, USP 50,000 USP units per 10 mL Vial Label - 50000 10mL container

This text provides information about a medication labeled as "Parin Sodium Injection, USP" with a concentration of 5,000 USP units per mL. It specifies that it is not intended for lock flush and is suitable for intravenous or subcutaneous use. The medication contains heparin sodium and sodium chloride as active ingredients and should be stored at a temperature between 20° to 25°C (68° to 77°F). Additionally, the product information indicates that the medication is manufactured in China for Northstar Rx LLC.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.