Product Images Tocilizumab-anoh
View Photos of Packaging, Labels & Appearance
- Figure 1 - tocilizumab 01
- Figure 2 - tocilizumab 02
- Figure A - tocilizumab 03
- Figure B - tocilizumab 04
- Figure C - tocilizumab 05
- Figure D - tocilizumab 06
- Figure E - tocilizumab 07
- Figure F - tocilizumab 08
- Figure G - tocilizumab 09
- Figure H - tocilizumab 10
- Figure I - tocilizumab 11
- Figure J - tocilizumab 12
- Figure K - tocilizumab 13
- Figure L - tocilizumab 14
- Figure M - tocilizumab 15
- Figure N - tocilizumab 16
- Figure A - tocilizumab 17
- Figure B - tocilizumab 18
- Figure C - tocilizumab 19
- Figure D - tocilizumab 20
- Figure E - tocilizumab 21
- Figure F - tocilizumab 22
- Figure G - tocilizumab 23
- Figure H - tocilizumab 24
- Figure I - tocilizumab 25
- Figure J - tocilizumab 26
- Figure K - tocilizumab 27
- Figure L - tocilizumab 28
- Figure M - tocilizumab 29
- Figure N - tocilizumab 30
- Figure O - tocilizumab 31
- Principal Display Panel - 80 mg/4 mL Vial Carton - tocilizumab 32
- Principal Display Panel - 200 mg/10 mL Vial Carton - tocilizumab 33
- Principal Display Panel - 400 mg/20 mL Vial Carton - tocilizumab 34
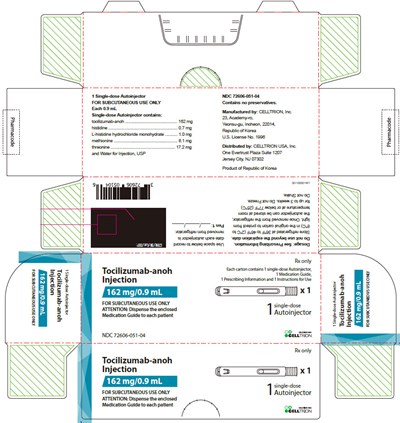
- Principal Display Panel - 162 mg/0.9 mL Autoinjector Carton - tocilizumab 35
Product Label Images
The following 35 images provide visual information about the product associated with Tocilizumab-anoh NDC 72606-051 by Celltrion Usa, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure A - tocilizumab 03

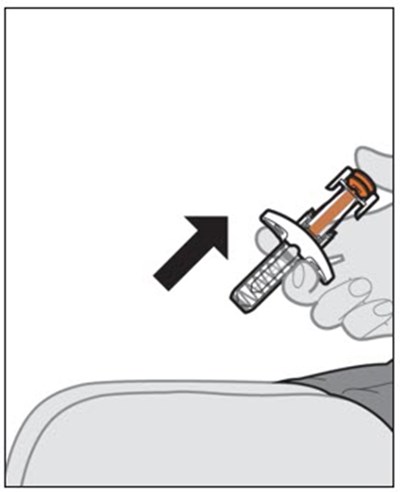
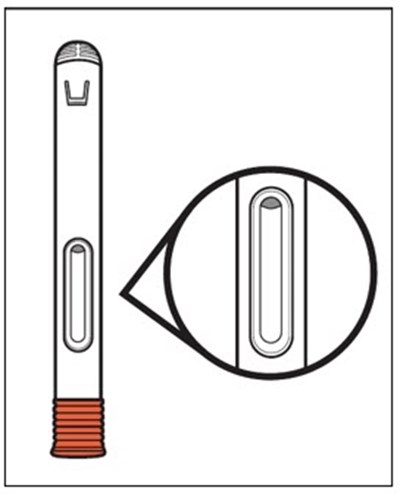
This text appears to be a list of items related to a medical device or tool. It includes terms such as "Plunger", "White", "Finger", "Flange", "Viewing", "Medicine Window", "Needle", "Guard", and "After Use". These items are likely components or features of a product that is used before and after use.*
Figure B - tocilizumab 04

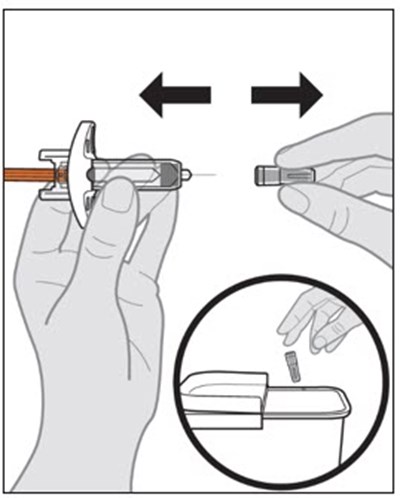
A description of the contents of a medical kit which includes a carton with a prefilled syringe, cotton ball or gauze, alcohol swab, adhesive bandage, and a FDA-cleared sharps disposal container.*
Figure A - tocilizumab 17

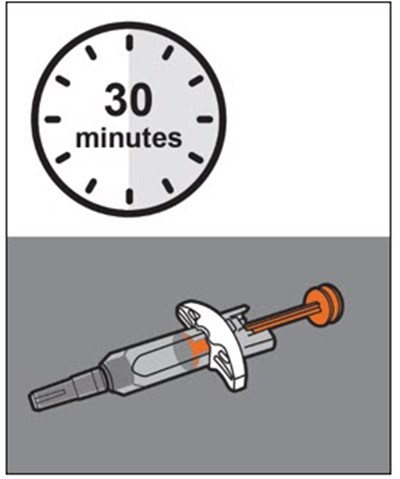
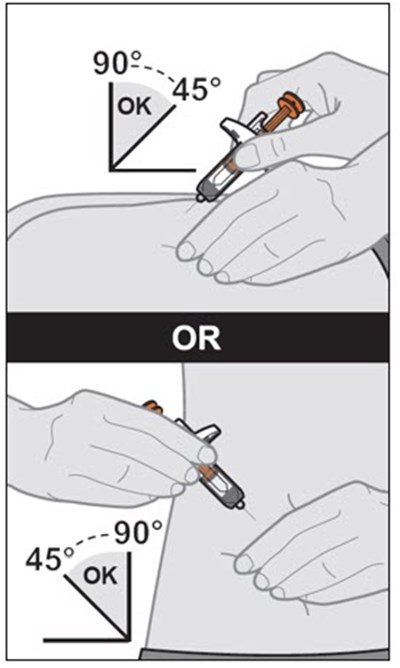
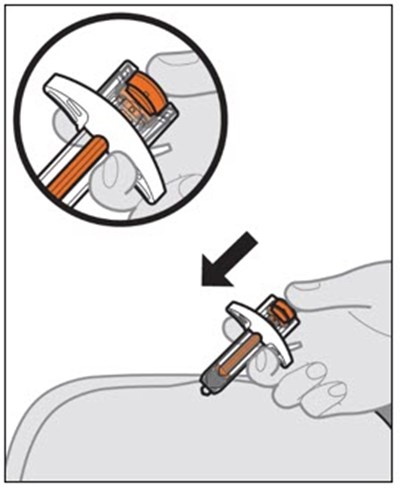
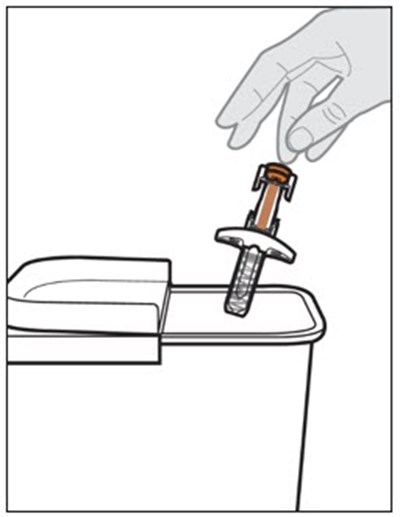
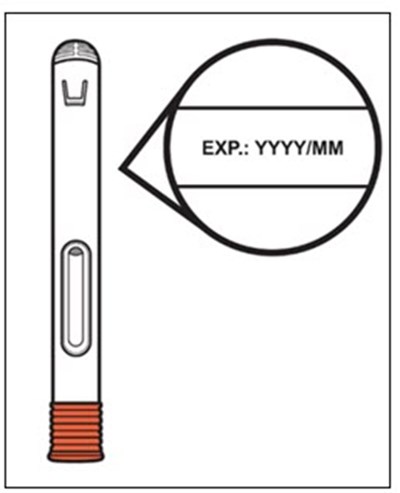
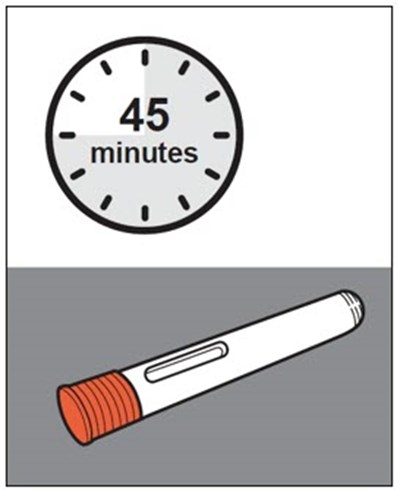
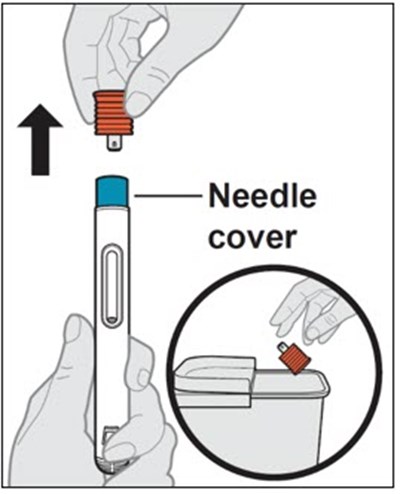
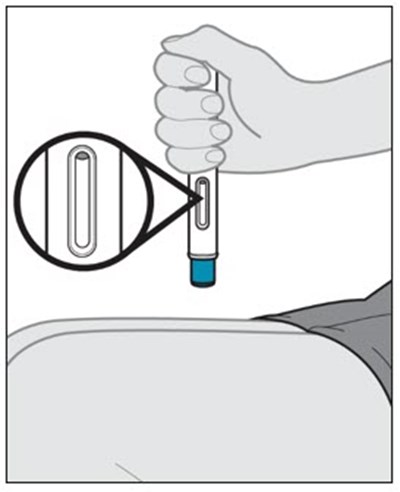
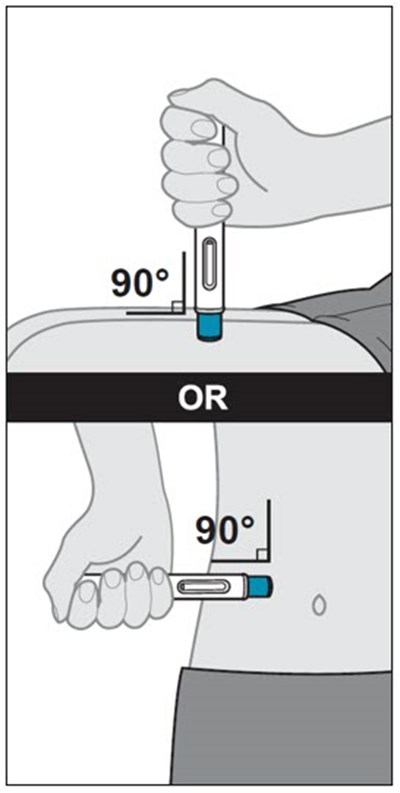
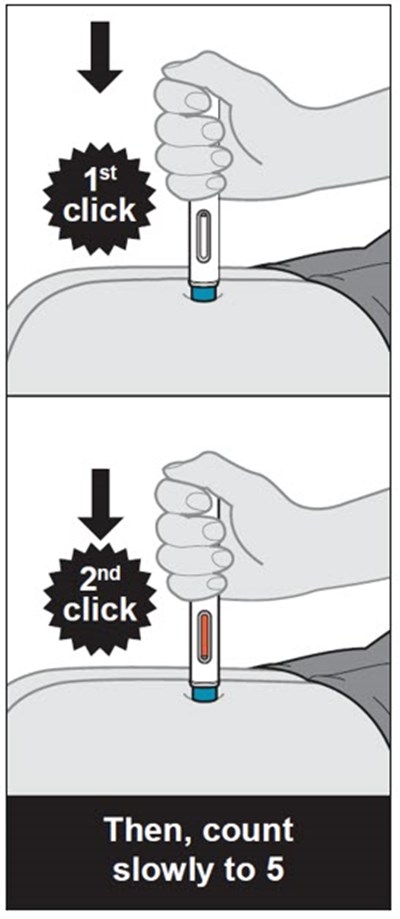
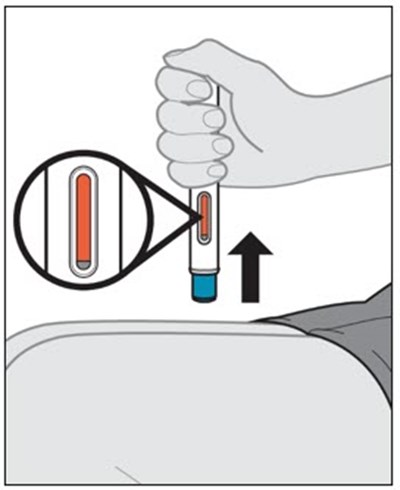
This text provides instructions and details about a medical device before and after use. It includes information about an expiration date, an orange indicator to show that the injection is complete, and a blue needle cover that is locked with the needle inside after use. The text seems to be a label or guide for using the device safely and effectively.*
Figure B - tocilizumab 18

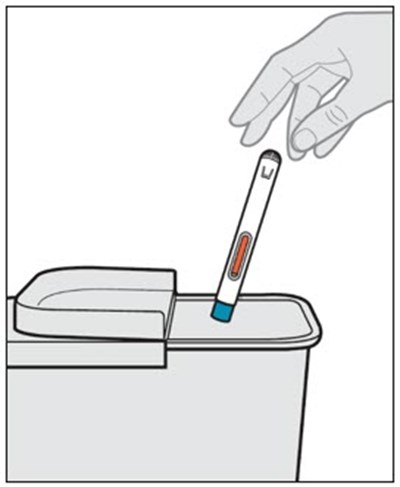
This text appears to be a list of the contents of a carton, which include cotton balls, an auto-injector, gauze, alcohol swab, adhesive bandage, and a sharps disposal container. The items mentioned seem to be for medical or health-related purposes.*
Principal Display Panel - 400 mg/20 mL Vial Carton - tocilizumab 34

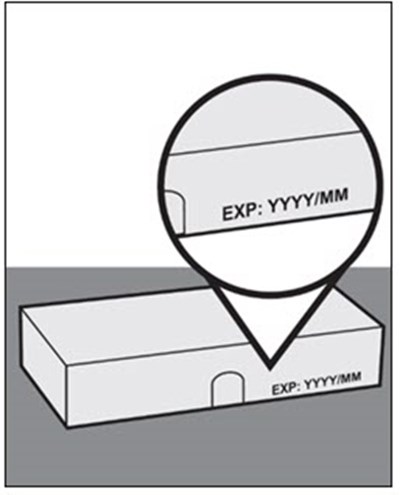
This is a description of Tocilizumab-anoh, an injection with a concentration of 400 mg/20 mL (20 mg/mL), intended for intravenous infusion after dilution. It comes in a single-dose vial which should be refrigerated at 2°C to 8°C (36°F to 46°F) to protect it from light. Each vial contains a solution with various components like tocilzumab-anoh, histidine, methionine, and more. The solution has a pH of 6.0 and does not contain any preservatives. The product is manufactured by CELLTRON, Inc in South Korea and distributed by CELLTRION USA, Inc in the USA. The dosage details are provided in the prescribing information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.