Product Images Fosaprepitant
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Fosaprepitant NDC 72606-569 by Celltrion Usa, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
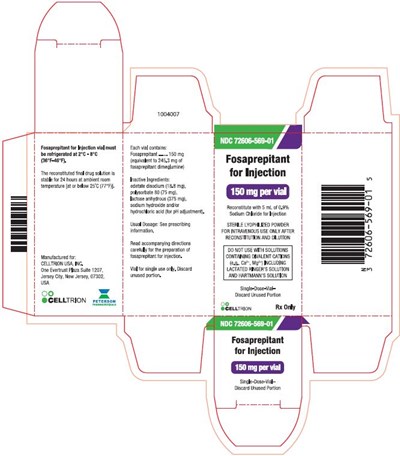
image description - 3fosaprepitantvial

This is a description of medication with the NDC code 72606-569-01, Fosaprepitant. The vial should be refrigerated between 2°C-8°C (36°F~46°F). The final drug solution is stable for 24 hours. The medication can be stored at ambient room temperature at or below 25°C (7°F). The recommended dosage is not mentioned. The medication comes in a single-dose vial with 150mg of lyophilized powder for intravenous use. Once reconstituted and diluted, the unused portion must be discarded. The manufacturer is Celltrion USA, Inc.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.