Product Images Pregabalin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 22 images provide visual information about the product associated with Pregabalin NDC 72658-0808 by Eskayef Pharmaceuticals Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure 8 - figure0008

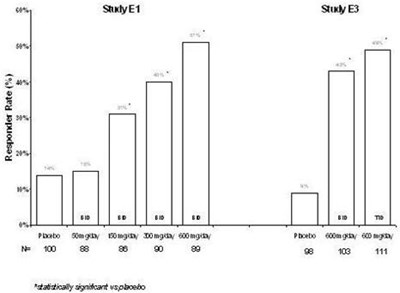
This appears to be a table displaying the responder rates for different dosages of a medication and a placebo. The percentages indicate the percentage of participants who responded positively to the treatment. The dosages tested were 2.5 mg/kg/day and 10 mg/kg/day. Participants who received the placebo had a responder rate of 22.6%. It is not possible to determine what the medication was for or any further details based on this limited information.*
figure 3 - figure003

This text is invalid and not readable. It seems to be a mix of errors, symbols, and letters that do not form a meaningful description.*
figure 11 - figure010
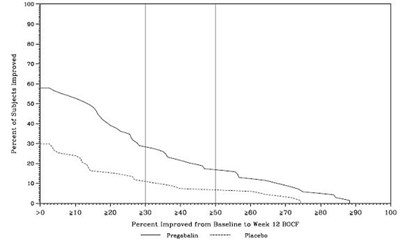
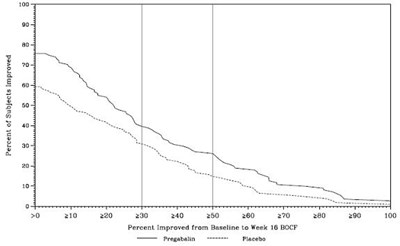
The text is a table showing the percentage of subjects who have improved from baseline to week 12 BOCF after taking Pregabalin. The percentages range from 50% to 100%, indicating high efficacy of the medication.*
Formula - formula001
This text presents a formula to calculate the glomerular filtration rate (GFR) based on the age, weight, and serum creatinine levels of a patient. The formula involves multiplying 140 minus the age of the patient in years by their weight in kilograms and then by their serum creatinine level in milligrams per deciliter. However, for female patients, the result is multiplied by 0.85.*
pregablin-100-60s-v2 - pregablin 100 60s v2

This is a prescription medication called Pregabalin EV Capsules. Each capsule contains 100mg of pregabalin and it should be stored at 20-25°C. The temperature range of 15-30°C is also permitted. Dispensing should be done in tight and child-resistant containers. The usual dosage and other important information are included in the prescribing information. The medication guide should be given to each patient. It is manufactured by Eskaysf Pharmaceuticals Limited in Bangladesh.*
pregablin-150-60s-v2 - pregablin 150 60s v2

This text is a label for Pregabalin capsules. It provides information on temperature limits and proper storage in tight and child-resistant containers. It also advises the pharmacist to dispense the medication guide to patients and refers to the accompanying prescribing information for dosage. The capsules contain 50 mg of Pregabalin, manufactured by Eskayef Pharmaceuticals in Bangladesh. The lot number and expiration date are also provided.*
pregablin-225-60s-v2 - pregablin 225 60s v2

This is a medication for which the recommended storage temperature ranges from 20 to 25 degrees Celsius. Excursions are allowed between 15 and 30 degrees Celsius. Each capsule contains 225mg of pregabalin USP. Dispense in tight child-resistant containers and provide the medication guide to each patient. The manufacturer is Eskayef Pharmaceuticals Limited, located in Bangladesh. No dosage information is provided.*
pregablin-25-60s-v2 - pregablin 25 60s v2

This is a medication called Pregabalin EV available in capsule form. Each capsule contains 25mg of Pregabalin, USP. The medication is supposed to be stored between 20° to 25° C, with excursions allowed between 15° to 30° C. The medication should be dispensed in a tight (USP), child-resistant container. Pharmacists should give out the accompanying medication guide to each patient. The usual dosage information is available in the accompanying prescribing information. The medication is manufactured by Eskaysf Pharmaceuticals Limited and the package contains 60 capsules.*
pregablin-300-60s-v2 - pregablin 300 60s v2

This appears to be a medication label for Pregabalin capsules produced by Eskayef Pharmaceuticals. The medication is to be stored at temperatures between 20°C to 25°C (68°F to 77°F) and is permitted to be stored between 15°C to 30°C (59°F to 86°F). The capsules come in child-resistant containers and the pharmacist is advised to dispense the accompanying medication guide to each patient. Each capsule contains 600mg of pregabalin, USP. The product has a GTIN number and an expiration date.*
pregabalin-50-60s-v2 - pregablin 50 60s v2

This is a medication guide for Pregabalin Capsules containing 50mg of pregabalin, USP. The medication is dispensed in child-resistant containers and should be stored at controlled room temperature. The usual dosage should be determined according to the accompanying prescribing information. The manufacturer is Eskayef Pharmaceuticals Limited, located in Bangladesh. The accompanying medication guide should be dispensed to each patient by the pharmacist. There is an NDC code and lot number provided, but there is no readable expiration date.*
pregabalin-75-60s-v2 - pregablin 75 60s v2

This is information for a medication called Pregabalin EV, which comes in capsule form. It should be stored at a temperature between 20-25°C and can tolerate excursions between 15-30°C. Each capsule contains 75mg of pregabalin. The manufacturer is Eskayef Pharmaceuticals Limited and the NDC code is 72658-0810-1. Further dosage information is provided in the accompanying prescribing guide, which should be given to each patient along with the medication.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.