Product Images Leuprolide Acetate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 15 images provide visual information about the product associated with Leuprolide Acetate NDC 72664-611 by Vgyaan Pharmaceuticals Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Carton - vial carton

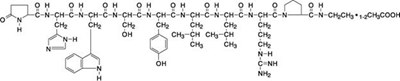
Leuprolide Acetate Injection is a prescription drug with a strength of 4mg/2.8mL. The medicine is for subcutaneous injection containing 1mg of leuprolide acetate in 0.2mL solution. The solution is sterile and contains sodium chloride to adjust tonicity, benzy| alcohol as a preservative and water for injection. The pH range of the solution is 4.0t06.0, and it may contain sodium hydroxide and/or acetic acid. The medication is stored below 77°F (26°C) and must not be frozen or exposed to light. The package insert has complete prescribing information and patient use instructions. Keep out of the reach of children. It is distributed by VGYAAN Pharmaceuticals LLC, Skillman, NJ, and made in the USA.*
Label - vial label

This is a pharmaceutical product labeled as NDC 72664-611-28 containing leupro' ide acetate with a usual dosage of 0.2 mL. It is recommended for subcutaneous injection once daily and should be stored below 77°F (25°C). It is important to protect the product from light and keep it out of the reach of children. The product is distributed by VGYAAN and made in the USA. The package insert should be consulted for more information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.