Product Images Losartan Potassium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Losartan Potassium NDC 72789-044 by Pd-rx Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
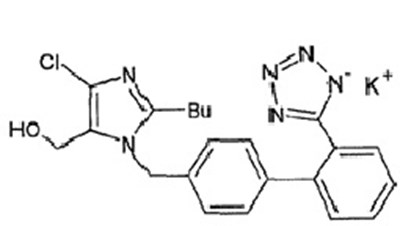
72789044 Label - 72789044

This is a medication label for Losartan Potassium USP tablets in 100mg. The label indicates that the medication should be discontinued when pregnancy is detected. It advises consumers to contact a doctor for medical advice on side effects and to report adverse reactions to the FDA. The label also provides instructions on dosage and warns against tearing the label.*
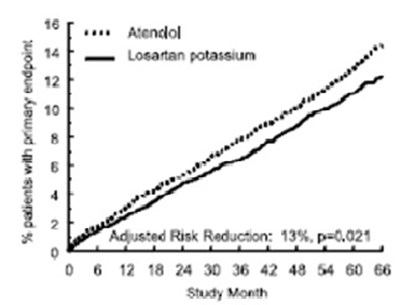
losartanfigure2 - losartanfigure2

This text represents a result summary of a study on the adjusted risk reduction for stroke or heart attack. The risk reduction achieved was 26% with a p-value of 0.001, indicating statistical significance. The graph shows the percentage of patients who experienced a first-line or fatal stroke or heart attack over a period of 66 months.*
losartanfigure4 - losartanfigure4
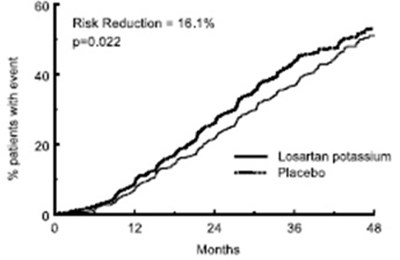
This text provides information about a measure called "Risk Reduction" that seems to be represented by the number "0022". It also shows the percentage of patents with an event associated with this measure, which is 18.1%. Additionally, it mentions the time period ("Months") in which this measure was evaluated, but there is not enough context to further describe it.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.