Product Images Sotalol Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Sotalol Hydrochloride NDC 72789-136 by Pd-rx Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
72789135 Label - 72789135

This is a medication containing Sotalol Hydrochloride. Each tablet has 160 mg. It is recommended to check usual dosage on the package insert. It should be stored in a light-resistant container, within controlled temperature conditions. It is marketed and packaged by PD-Rx Pharmaceuticals and manufactured by Oxford Pharmaceuticals. The bottle contains 100 tablets. The product's expiration date is 04/2022 and the LOT number is J20A35.*
72789136 Label - 72789136

Each tablet contains Sotalol Hydrochloride USP at a strength of 240 mg. It is manufactured by Oxford Pharmaceuticals and marketed by PD-Rx Pharmaceuticals, Inc. The product comes in a bottle containing 100 tablets and should be stored in a cool, dry place. The standard dosage should be referred to by consulting the package insert. Other details, such as GTIN, LOT, and SNO codes are also available.*
72789137 Label - 72789137

Each tablet contains 80mg of Sotalol Hydrochloride, manufactured by Oxford Pharmaceuticals LLC and marketed by PD-Rx Pharmaceuticals Inc. The tablets should be stored at a controlled temperature of 20° to 25°C (68° to 77°F). The usual dosage is to be determined by following the instructions given in the package insert. The product comes in a pack of 100 tablets and is identified by a GTIN of 00372789137018 and other identification numbers.*
72789138 Label - 72789138

Each tablet contains 120 mg of Sotalol Hydrochloride, indicated for the treatment of heart rhythm disorders. The tablets are packaged in a tight and light-resistant container and must be stored at a controlled room temperature. The usual dosage information is available in the package insert. The product is manufactured by PD-Rx Pharmaceuticals, Inc and packaged by Oxford Pharmaceuticals, LLC. The product is available in a bottle containing 100 tablets. The NDC number is 72789-138-01, and the expiration date is 04/2022.*
Figure 2 - figure2
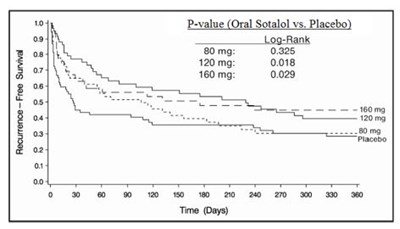
The text shows the p-values indicating the comparison of recurrence-free survival between oral Sotalol and Placebo for different dosages. The numbers represent the different dosages and the corresponding p-values: 80mg-0325, 120mg-0018, 160mg-0029. No other information is available to provide further context.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


