Product Images Desmopressin Acetate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Desmopressin Acetate NDC 72843-440 by Ubi Pharma Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10mL carton label - 10ml carton label
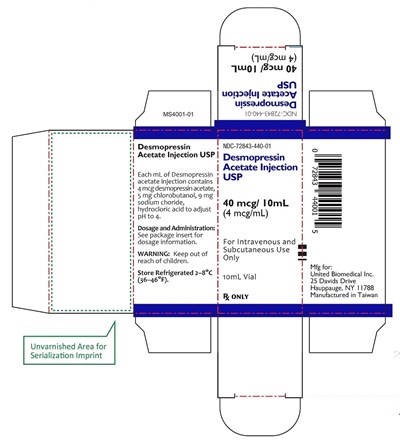
Desmopressin Acetate Injection USP is a medication provided in 10mL vials containing 4mcg desmopressin acetate per mL, also including 5mg chlorobutanol, 9mg sodium chloride, and hydrochloric acid. The product is intended for intravenous or subcutaneous administration and should be stored refrigerated between 2°C-8°C (36-46°F). The package suggests reference to the insert for dosage information and warns to keep the product out of reach of children. The medication is manufactured in Taiwan by United Biomedical Inc.*
10mL container label - 10ml container label

Desmopressin Acetate Injection is a medication that is administered intravenously and subcutaneously. This medication is available in a 10 ml vial with a concentration of 4 megiml. Please refer to the package insert for dosage information. Keep away from children and store refrigerated between 2-8°C (36-46°F). This medication is manufactured by United Biomedical Inc. at their Hauppauge, NY facility.*
1mL carton label - 1ml carton label
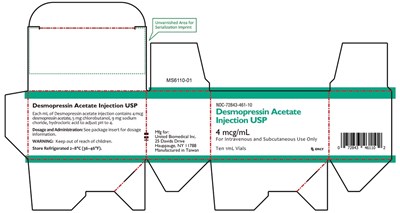
Desmopressin Acetate Injection USP is a medication used for Intravenous and Subcutaneous use only. Each mL of the injection contains 4 mg of Desmopressin acetate, 5 mg of chiorobutanal, 10 mg of sodium chloride, and hydrochloric acid to adjust pH to 4. Dosage and Administration instructions are provided in the package insert. The medication needs to be stored refrigerated at 2-8°C (36-46°F) and kept out of reach of children. The product is manufactured in Taiwan by United Biomedical Inc. This specific packaging contains ten 1L vials.*
1mL container label - 1ml container label

This is a medication package insert for Desmopressin Acetate in Injection USP format, with a concentration of 4 mcg/mL. It provides instructions for dosage and administration, stating that the medication should be kept out of reach of children and stored in a refrigerated environment. The medication is intended for intravenous and subcutaneous use only. The manufacturer is United Biomedical Inc., located in Hauppauge, NY. No further information about the medication's uses or effects is provided.*
desmopressin structure - desmopressin structure

The text appears to be a chemical formula of a peptide with the sequence "C-Tyr-Phe-Gln-Asn-Cys-Pro-D-Arg-Gly-NHz" and a molecular weight of 3345. It may be useful in the field of biochemistry and pharmaceuticals.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.