Product Images Levothyroxine Sodium
View Photos of Packaging, Labels & Appearance
- 100 mcg - 1000 count - 100 mcg 1000s
- 100 mcg - 90 count - 100 mcg 90s
- 112 mcg - 1000 count - 112 mcg 1000s
- 112 mcg - 90 count - 112 mcg 90s
- 125 mcg - 1000 count - 125 mcg 1000s
- 125 mcg - 90 count - 125 mcg 90s
- 137 mcg - 1000 count - 137 mcg 1000s
- 137 mcg - 90 count - 137 mcg 90s
- 150 mcg - 1000 count - 150 mcg 1000s
- 150 mcg - 90 count - 150 mcg 90s
- 175 mcg - 1000 count - 175 mcg 1000s
- 175 mcg - 90 count - 175 mcg 90s
- 200 mcg - 1000 count - 200 mcg 1000s
- 200 mcg - 90 count - 200 mcg 90s
- 25 mcg - 1000 count - 25 mcg 1000s
- 25 mcg - 90 count - 25 mcg 90s
- 300 mcg - 1000 count - 300 mcg 1000s
- 300 mcg - 90 count - 300 mcg 90s
- 50 mcg - 1000 count - 50 mcg 1000s
- 50 mcg - 90 count - 50 mcg 90s
- 75 mcg - 1000 count - 75 mcg 1000s
- 75 mcg - 90 count - 75 mcg 90s
- 88 mcg - 1000 count - 88 mcg 1000s
- 88 mcg - 90 count - 88 mcg 90s
- Structure - Structure
- Table1 - Table1
- table 2 - Table2
- Table3 - Table3
- Table4 - Table4
- Table5 - Table5
- Table6 - Table6
- Table7 - Table7
Product Label Images
The following 32 images provide visual information about the product associated with Levothyroxine Sodium NDC 72865-237 by Xlcare Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
100 mcg - 1000 count - 100 mcg 1000s

Levothyroxine Sodium Tablets, with a strength of 100mcg, are manufactured by Ascend Pharmaceutical, and distributed under the brand name XLcare by XLCare Pharmaceuticals. The medicine should be protected from both moisture and light. The full prescribing information can be found on the packaging.*
100 mcg - 90 count - 100 mcg 90s

This is a description of a medication named Levothyroxine Sodium Tablets, USP available in a pack of 90 tablets with a strength of 100 meg. The NDC number of the medication is 72865-240-90. The medication is for prescription use only. The other text in the provided is not readable.*
112 mcg - 1000 count - 112 mcg 1000s

This text appears to be a label for a medication called "Levothyroxine Sodium Tablets, USP" with a dosage and administration instructions that are not available in this text. The medication is manufactured by Ascent Pharmaceutical and sold by XLCare Pharmaceuticas. The label also contains a manufacturer's warning about protecting the medication from moisture and light.*
112 mcg - 90 count - 112 mcg 90s

Levothyroxine Sodium Tablets, USP with NDC 72865-241-90 is a prescription-only medication for treating low thyroid hormone. It comes with 90 tablets in a bottle. The other text includes manufacturing and distribution information.*
125 mcg - 1000 count - 125 mcg 1000s

This is a prescription drug called Levothyroxine Sodium Tablets manufactured by Ascent Pharmaceuticals for XLCare Pharmaceuticals. It comes in a bottle containing 1000 tablets and NDC 72865-242-10. It should be protected from moisture and light and the full prescribing information should be read before use. The address of XLCare Pharmaceuticals is listed as 242 South Culver Street, Suite 202, Lawrenceville, GA 30045.*
125 mcg - 90 count - 125 mcg 90s

This is a product packaging of Levothyroxine Sodium Tablets, USP with NDC code 72865-242-90. There are 90 tablets in the bottle and it is to be used under prescription only. The rest of the text consists of some unclear characters and therefore cannot be interpreted.*
137 mcg - 1000 count - 137 mcg 1000s

XLcare is a brand that produces Levothyroxine Sodium Tablets, USP of 137mcg (0.137mg). The tablets come in a bottle of 1000 tablets and are prescribed medication. The medicine should be stored in a tightly closed container in a controlled room temperature, protected from moisture and light. The prescription information brochure should be read for information on dosage and application. The medication is manufactured by Ascent Pramacaiticas, INC located in Centralsip, NY 11722, and distributed by XLCare Pharmaceuticals, INC located in Lawrenceville, GA 30046.*
137 mcg - 90 count - 137 mcg 90s

Levothyroxine Sodium Tablets, USP with a strength of 137 mcg (0137 mg) and 90 tablets per bottle. The NDC number is 72865-243-90 and it is only available with a prescription. The rest of the text contains irrelevant information and incomplete words, making it not useful for description.*
150 mcg - 1000 count - 150 mcg 1000s

Levothyroxine Sodium Tablets, USP is a medication manufactured by XLCare Pharmaceuticals, Inc. It is used for the treatment of certain thyroid disorders and comes in a bulk package of 1000 tablets with the strength of 150 mcg (0.15 mg). The medication should be protected from moisture and light. The full prescribing information should be consulted before use. The NDC number for the medication is 72865-244-10, and it can be dispensed only by prescription.*
150 mcg - 90 count - 150 mcg 90s

This appears to be a prescription label for 90 tablets of Levothyroxine Sodium Tablets, USP at 150 mcg per tablet. The label includes a product code (WG 72865-244-00) and a lot number (20050), as well as a pharmacy address (S St 22).*
175 mcg - 1000 count - 175 mcg 1000s

Levothyroxine Sodium Tablets, marketed by XLcare Pharmaceuticals, are used to treat hypothyroidism (underactive thyroid) and other thyroid conditions. The tablets contain 0.175 mg of the active ingredient and should be protected from moisture and light. The full prescribing information for this medication can be found on the package insert with NDC number 72865-245-10. For any queries or to order, contact XLCare Pharmaceuticals at their Lawrenceville, GA address or call them on 01 592,6982.*
175 mcg - 90 count - 175 mcg 90s

This is a description of a medication named Levothyroxine Sodium Tablets, USP that are packaged in a bottle containing 90 tablets. The NDC (National Drug Code) number is 72865-245-90. The medication is available only with a prescription. The remaining text appears to be miscellaneous information and therefore could not be interpreted.*
200 mcg - 1000 count - 200 mcg 1000s

Levothyroxine Sodium Tablets, USP is a medication used to treat an underactive thyroid (hypothyroidism). The medication comes in tablets of 200 mcg and is manufactured by Ascent Pharmaceuticals. It is distributed by XLCare Pharmaceuticals, Inc. The tablets should be protected from moisture and light. For more information about the medication, refer to the prescribing information.*
200 mcg - 90 count - 200 mcg 90s

Levothyroxine Sodium Tablets, USP is a prescription medicine containing 200mcg (0.2mg) of the active ingredient and packaged in a bottle containing 90 tablets. The NDC number for the drug is 72865-246-90 and the package is marked "RxOnly". The remaining text contains information related to the manufacturer's address, including the city, state, and zip code. The product is intended for human consumption.*
25 mcg - 1000 count - 25 mcg 1000s

This is a prescription drug information for XLecare's Levothyroxine Sodium Tablets, a dispersive medication used as a hormone replacement therapy for those with an underactive thyroid gland. It is manufactured by Ascent Pharmaceutical and distributed by XLCare Pharmaceuticals. The packaging contains 1000 RX only tablets and should be protected from moisture and light. No useful information can be obtained from the error at the end.*
300 mcg - 1000 count - 300 mcg 1000s

Levothyroxine is a prescription medicine used for the treatment of thyroid hormone deficiency. The medicine is in the form of sodium tablets and should be protected from moisture and light. The tablets are manufactured by Ascent Pharmaceutical and distributed by XLCare Pharmaceuticals in the dosage form of 1000 tablets. The label contains information about the manufacturer, Centrealsip, NY 11722, and the distributor, Lawrenceville, GA 30045. The product is dispensed under NDC code 72865-247-10. The full prescribing information is available for this medication.*
300 mcg - 90 count - 300 mcg 90s

This is an incomplete description and appears to be a list of random words and phrases. It is not readable and therefore not available for generating useful content.*
50 mcg - 1000 count - 50 mcg 1000s

Levothyroxine Sodium Tablets, USP is a prescription medication manufactured by Ascent Pharmaceuticals, Inc. and distributed by XLCare Pharmaceuticals, Inc. It is used to treat hypothyroidism and other thyroid problems and must be taken following the dosage and administration instructions provided in the full prescribing information. The product comes in a bottle of 1000 tablets and should be protected from moisture and light.*
50 mcg - 90 count - 50 mcg 90s

Levothyroxine Sodium Tablets, USP with strength 50mcg and package size of 90 tablets from XiCare. This is a prescription-only drug. The other text consists of codes and addresses.*
75 mcg - 1000 count - 75 mcg 1000s

Levothyroxine Sodium Tablets are a pharmaceutical product manufactured by Ascent Pharmaceuticals. These tablets are used to treat conditions related to an underactive thyroid gland. One bottle of the product contains 1000 tablets and the National Drug Code (NDC) for this product is 72865-238-10. The manufacturer recommends storing the tablets in a cool, dry place. For full prescribing information, consult the label. The tablets are sold by XLCare Pharmaceuticals and should only be used with a prescription from a licensed healthcare provider.*
88 mcg - 1000 count - 88 mcg 1000s

This appears to be the partial text of a drug prescription. The drug mentioned is "Levothyroxine Sodium Tablets, USP". It also provides information about protecting the medicine from moisture and light. Additionally, there are some details about the manufacturer and distributor of the medicine.*
88 mcg - 90 count - 88 mcg 90s

This is a description of a medication called Levothyroxine Sodium Tablets, USP with dosage strength of 88mcg in a pack of 90 tablets. The medication requires a prescription and the NDC number is 72865-239-90. The remaining characters do not provide any additional useful information.*
Table1 - Table1
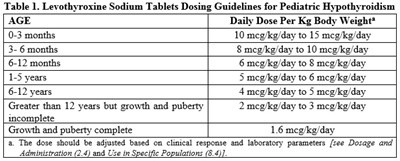
This text provides dosing guidelines for pediatric hypothyroidism using Levothyroxine Sodium Tablets. The daily dose per kg body weight varies based on the age of the patient, ranging from 10-15 meg/kg/day for patients aged 0-3 months, to 1.6 meg/kg/day for patients with complete growth and puberty. The dose should be adjusted based on clinical response and laboratory parameters.*
table 2 - Table2
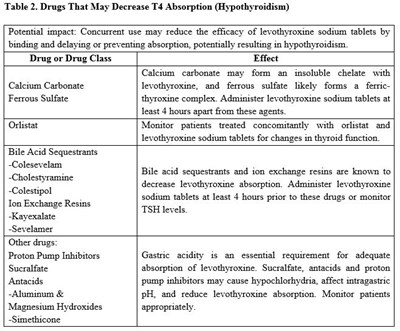
This is a table containing a list of drugs and drug classes that may decrease the absorption of levothyroxine sodium fablets, potentially leading to hypothyroidism. The drugs include Calcium Carbonate, Ferrous Sulfate, Bile Acid Sequestrants, Ton Exchange Resins, Proton Pump Inhibitors, Sucralfate, and Antacids with Aluminum & Magnesium Hydroxides and Simethicone. Patients using these drugs should consult a doctor and follow the recommended dosages to avoid reducing the efficacy of levothyroxine sodium tablets.*
Table3 - Table3

This is a table showing drugs that may affect the transport of T4 and T3 in serum without affecting the concentration of FT4 or causing thyroid dysfunction. The drugs mentioned can increase or decrease serum thyroxine-binding globulin concentration, and some may cause protein-binding site displacement. The use of these drugs with levothyroxine sodium tablets may cause an initial transient increase in FT4 followed by a decrease in serum T4, but normal FT4 and TSH concentrations are usually maintained. Salicylates may inhibit the binding of T4 and T3 to TBG and transthyretin, which can lead to an initial increase in serum FT4 followed by a return to normal levels with sustained therapeutic serum salicylate concentrations. Some drugs, such as phenytoin and carbamazepine, can reduce the serum protein binding of levothyroxine and may cause reduced total and free T4 levels, but most patients remain clinically euthyroid with normal serum TSH levels. Close monitoring of thyroid hormone parameters is recommended.*
Table4 - Table4

This text provides a table of drugs that may affect the metabolism of levothyroxine in the liver, resulting in increased requirements for levothyroxine sodium tablets. The table includes the drug or drug class and its effect on thyroid hormone levels. Phenobarbital and rifampin are listed as drugs that can increase the metabolism of levothyroxine. Barbiturates may also affect thyroid status if added or withdrawn from patients being treated for hypothyroidism.*
Table5 - Table5
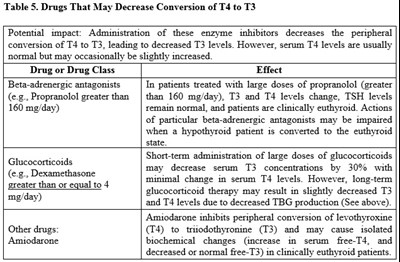
This text provides a table of drugs that can potentially decrease the conversion of T4 to T3, leading to lower levels of T3. It also discusses the effects of short and long-term administration of glucocorticoids on T3 and T4 levels. The text warns that patients treated with particular beta-adrenergic antagonists should be carefully monitored if undergoing a conversion to a euthyroid state. Furthermore, it mentions the impact of Amiodarone on the conversion of T4 to T3, which can cause specific biochemical changes in clinically euthyroid patients.*
Table6 - Table6
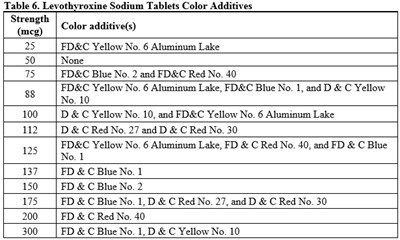
This text shows a table of color additives used in Levothyroxine Sodium Tablets for various strengths. Different color additives like FD&C Yellow No. 6 Aluminum Lake, FD&C Blue No. 2, FD&C Red No. 40, D&C Yellow No. 10, and D&C Red No. 27 are used with varying strengths of Levothyroxine Sodium Tablets.*
Table7 - Table7

Table 7 shows pharmacokinetic parameters in euthyroid patients for two thyroid hormones: Levothyroxine (T4) and Liothyronine (T3). The table lists their biological potency, half-life, and protein binding ratio. The biological potency of T4 is between 10-20 times greater than that of Thyroglobulin. The half-life of T4 is about 67 days, whereas that of T3 is much shorter- only 1 day. The protein binding ratio includes TBG, TBPA, and TBA. Finally, the note at the end of the table indicates that the half-life of these hormones can vary depending on thyroid levels, with 3 to 4 days during hyperthyroidism and 9 to 10 days during hypothyroidism.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


