FDA Label for Hand Sanitizer
View Indications, Usage & Precautions
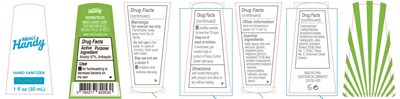
Hand Sanitizer Product Label
The following document was submitted to the FDA by the labeler of this product Merci Handy Corporation. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Otc - Active Ingredient
Active ingredient
Alcohol 67%
Otc - Purpose
Purpose
Antiseptic
Indications & Usage
Uses
for handwashing to decrease bacteria on the skin.
Warnings
Warnings
For external use only. Flammable, keep away from fire or flame.
Do not use in the eyes.In case of contact, flush eyes with water.
Otc - Stop Use
Stop use and ask a doctor if
- irritation and redness develop.
- condition persists for more than 72 hours
Otc - Keep Out Of Reach Of Children
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Dosage & Administration
Directions
wet hand thoroughly with product and allow to dry without wiping
Storage And Handling
Other information
store at a temperature below 110 F(43 C)
Inactive Ingredient
Inactive ingredients
water(aqua), aloe vera leaf juice, glycerin, propylene glycol, fragrance(parfum), acrylates/C30-10 alkyl acrylate crosspolymer, aminomethyl propanol, mannitol, microcrystalline cellulose, sucrose, corn(zea mays) starch, hydroxpropyl methyl cellulose, tocopheryl acetate, denatonium benzonate, maltodextrin, citral, geraniol, limonene, linalool, FD&C Blue No.1, FD&C Yellow No.5, Chromium Oxide Greens.
Package Label.Principal Display Panel
* Please review the disclaimer below.