Product Images Sildenafil Citrate
View Photos of Packaging, Labels & Appearance
- PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label - sildenafil tablets usp 20mg 1000 label
- PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label - sildenafil tablets usp 20mg 90 label
- Graph - sildenafil tablets usp 20mg figure 06
- Graph - sildenafil tablets usp 20mg figure 07
- Graph - sildenafil tablets usp 20mg figure 08
- Graph - sildenafil tablets usp 20mg figure 09
- Graph - sildenafil tablets usp 20mg figure 10
- Graph - sildenafil tablets usp 20mg figure 11
- Graph - sildenafil tablets usp 20mg figure 12
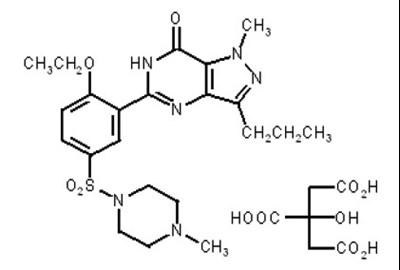
- Structure Formula - sildenafil tablets usp 20mg structure
Product Label Images
The following 10 images provide visual information about the product associated with Sildenafil Citrate NDC 72888-018 by Advagen Pharma Ltd, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label - sildenafil tablets usp 20mg 1000 label

This is a medication description for Sildenafil tablets. Each tablet contains 20mg of Sildenafil citrate USP. The usual dosage information can be found on the prescribing information. The tablets should be stored at controlled room temperature and dispensed in a tight container. The medication is only for adult use and should be kept out of the reach of children. More patient information can be found on the website. This is a prescription medication with 1,000 tablets per container. It is distributed by Advagen Pharma Limited and manufactured by Rubicon Research Private Limited in India.*
PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label - sildenafil tablets usp 20mg 90 label

Each tablet of the medicine contains 20 mg sildenafil citrate USP. The patient information sheet is available on the website mentioned. Look at the accompanying prescribing information for the usual dosage. The medicine is distributed by Advagen Pharma Ltd. It should be stored below 25°C and away from children's reach. The medicine is manufactured by Rubicon Research Private Limited, Ambernath, India. The details regarding the manufacturing license number are also given. However, it is not clear what the overprinting area is.*
Graph - sildenafil tablets usp 20mg figure 07
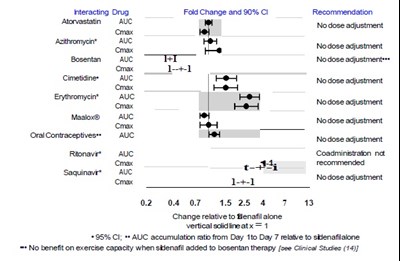
This text provides recommendations for nodose adjustment of various drugs, such as Acitomycin, Bosentan, Cimetidines, Evomyeint, Maslox, Oral Contraceptives, Sapionis, along with their respective AUC and Cmax values. The text also mentions the interaction of drug fold change and 90% CI recommendations. Additionally, there is a reference made to change relative to Sildenafil alone, with a vertical solid line indicating 1. The text also mentions that there is no benefit on exercise capacity when Sildenafil is added to Bosentan therapy.*
Graph - sildenafil tablets usp 20mg figure 08
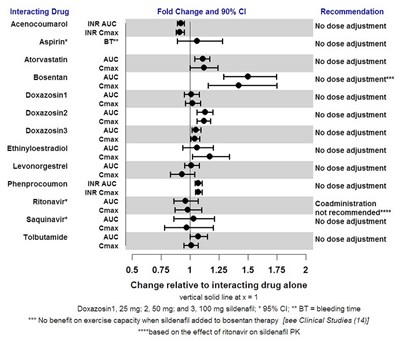
This is a drug interaction table showing the interaction of Acenocoumarol with other drugs such as Aspirin, Atorvastatin, Bosentan, Ethinyloestradiol, among others. The table provides recommendations for the fold changes and 90% CI for INR AUC, Cmax, and BT. It also indicates when co-administration is not recommended, such as the case of ritonavir with sildenafil PK. The table also shows that sildenafil has no benefit on exercise capacity when added to bosentan therapy.*
Graph - sildenafil tablets usp 20mg figure 09
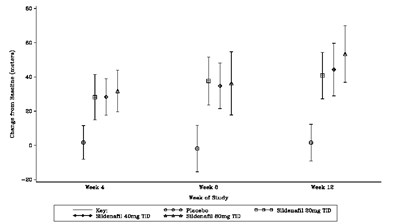
This is a table showing the change from baseline (in meters) for different doses of Sildenafil (a medication used to treat erectile dysfunction) in a 12-week study. The doses were placebo, 20mg TID, 40mg TID, and 80mg TID. The results were measured at weeks 4, 8, and 12.*
Graph - sildenafil tablets usp 20mg figure 10
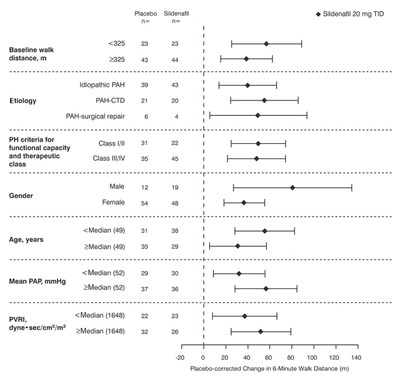
This appears to be a medical report assessing the effectiveness of Sildenafil (also known as Sidenafi) for treating Idiopathic Pulmonary Arterial Hypertension (IPAH). The report includes data on the patients' baseline walk distance, age, gender, and mean PAP (pulmonary artery pressure) before and after treatment with Sildenafil. The main measure of treatment effectiveness appears to be the change in 6-minute walk distance after treatment, which is graphed at the end of the report.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.