Product Images Yutrepia
View Photos of Packaging, Labels & Appearance
- Structural Formula - image 01
- Figure 1 - image 02
- Figure 2 - image 03
- Figure 3 - image 04
- Figure 4 - image 05
- Figure 5 - image 06
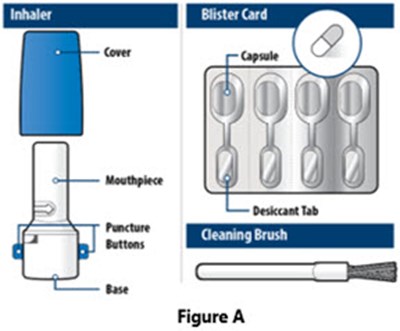
- Figure A - IFU - image 07
- Figure B - IFU - image 08
- Figure C - IFU - image 09
- Figure D - IFU - image 10
- Figure E - IFU - image 11
- Figure F - IFU - image 12
- Figure G - IFU - image 13
- Figure H - IFU - image 14
- Figure i - IFU - image 15
- Figure J - IFU - image 16
- Figure K - IFU - image 17
- Figure L - IFU - image 18
- Figure M - IFU - image 19
- Figure N - IFU - image 20
- Figure O - IFU - image 21
- Figure P - IFU - image 22
- Figure Q - IFU - image 23
- Figure R - IFU - image 24
- Figure S - IFU - image 25
- When dosing with more than one capsule... - image 26
- Figure T - IFU - image 27
- Figure U - IFU - image 28
- Figure V - IFU - image 29
- Figure W - IFU - image 30
- Figure X - IFU - image 31
- Principal Display Panel - 26.5 mcg Carton - image 32
- Principal Display Panel - 53 mcg Carton - image 33
- Principal Display Panel - 79.5 mcg Carton - image 34
- Principal Display Panel - 106 mcg Carton - image 35
Product Label Images
The following 35 images provide visual information about the product associated with Yutrepia NDC 72964-013 by Liquidia Technologies, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
When dosing with more than one capsule... - image 26

This text provides instructions on dosing with more than one capsule when the prescribed dose is 132.5 mcg or larger. It explains that if multiple capsules are needed to complete the dose, the steps from 4 to 13 should be repeated for each additional capsule. Additionally, it mentions that the order in which the capsules are dosed does not matter regardless of their strength.*
Principal Display Panel - 79.5 mcg Carton - image 34

This document appears to be a pharmaceutical label for an inhalation powder medication called Yutreplo. It provides instructions for use, dosage information, storage guidelines, and warnings to keep the medication out of reach of children. The label also includes lot number and copyright information for Liquidia, the company that manufactures the product.*
Principal Display Panel - 106 mcg Carton - image 35

This is a prescription label for Yutrepro inhalation powder containing treprostinil. The recommended dosage instructions and storage conditions are provided on the label. It is important to keep the medication out of reach of children and not remove the capsules from the blister pack until ready to use. Be sure to follow the prescribing information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.