Product Images Tyrvaya
View Photos of Packaging, Labels & Appearance
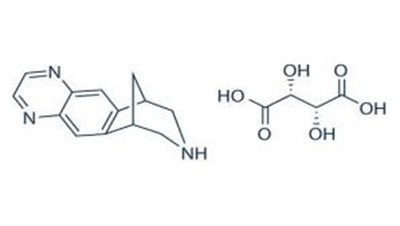
- The chemical structure - varenicline 01
- Tyrvaya Nasal Spray - varenicline 02
- Remove clip/cap - varenicline 03
- Step 2. Hold the nasal spray upright and away from your face. Place one finger on each side of the base of the nasal applicator, and place your thumb underneath the bottle. - varenicline 04
- Press 7 times - varenicline 05
- Step 1. Blow nose to clear nostrils if needed. - varenicline 06
- Remove cap/clip - varenicline 07
- Step 3. Hold the nasal spray upright. Place one finger on each side of the base of the nasal applicator and your thumb underneath the bottle. - varenicline 08
- Step 4. Tilt your head back slightly. - varenicline 09
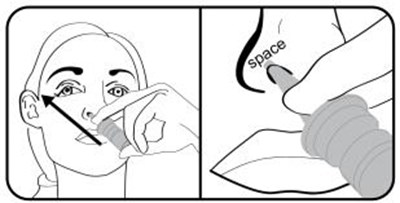
- Step 5. Insert the nasal applicator into the left or right nostril. Tilt the nasal applicator and aim the tip towards the top of the ear on the same side. DO NOT press the tip of nasal applicator against the wall of the inside of your nose. Allow for a space between the tip of nasal applicator and the wall of the inside of your nose for proper dose administration. - varenicline 0a
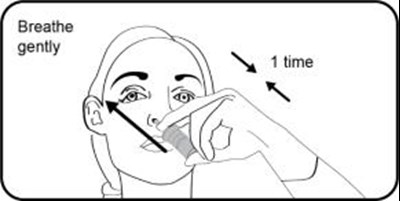
- Step 6. Place tongue to the roof of the mouth and breathe gently while spraying 1 (one) time. Repeat administration in the other nostril. Wait 2 (two) or 3 (three) minutes before blowing nose if needed. - varenicline 0b
- Step 7. Wipe the nasal applicator with a clean tissue. - varenicline 0c
- Step 8. Replace clip and cap. - varenicline 0d
- DiagramDescription automatically generated - varenicline 0e
- TextDescription automatically generated - varenicline 0f
- DiagramDescription automatically generated - varenicline 10
- Graphical user interface, text, applicationDescription automatically generated with medium confidence - varenicline 11
Product Label Images
The following 17 images provide visual information about the product associated with Tyrvaya NDC 73521-030 by Oyster Point Pharma, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Step 2. Hold the nasal spray upright and away from your face. Place one finger on each side of the base of the nasal applicator, and place your thumb underneath the bottle. - varenicline 04
Remove cap/clip - varenicline 07

The text suggests an action in a set of instructions where the user must remove a cap and/or a clip from a certain object. The object and the purpose of the action are not provided.*
Step 3. Hold the nasal spray upright. Place one finger on each side of the base of the nasal applicator and your thumb underneath the bottle. - varenicline 08

Step 5. Insert the nasal applicator into the left or right nostril. Tilt the nasal applicator and aim the tip towards the top of the ear on the same side. DO NOT press the tip of nasal applicator against the wall of the inside of your nose. Allow for a space between the tip of nasal applicator and the wall of the inside of your nose for proper dose administration. - varenicline 0a
Step 6. Place tongue to the roof of the mouth and breathe gently while spraying 1 (one) time. Repeat administration in the other nostril. Wait 2 (two) or 3 (three) minutes before blowing nose if needed. - varenicline 0b
DiagramDescription automatically generated - varenicline 0e
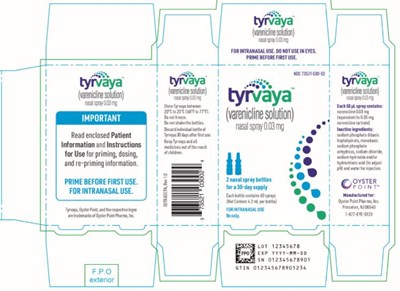
This text seems to be a label of a nasal spray medication. It gives instructions for priming the spray before first use and specifies that it is for intranasal use. It also provides the lot number and expiration year of the medication. The label contains an FPO mark and a logo for Oyster Point Pharma.*
TextDescription automatically generated - varenicline 0f

Each bottle of Tyrvaya spray contains 60 sprays with varenicline tartrate as the active ingredient. The spray is used for smoking cessation purposes. It is recommended to keep the spray between 20°C to 25°C (66°F to 77°F) and not freeze it. The bottle should be discarded after 30 days from the first use. For further information, see the package insert. The product is manufactured by M. forOyste Pint Parms, Inc.*
DiagramDescription automatically generated - varenicline 10
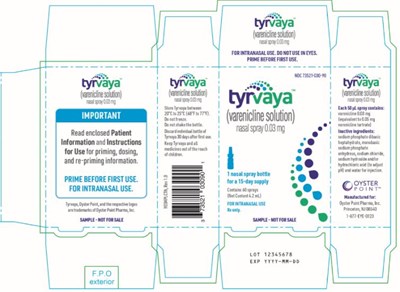
This is a package insert label for a nasal spray which provides instructions for priming, dosing, and re-priming. It is emphasized to prime the spray before first use and avoid using it in the eyes. The label includes the product name, lot number, and expiration date. However, it does not mention the name of the medication, the manufacturer's name, or any dosage instructions, thus, further information might be needed to identify the product.*
Graphical user interface, text, applicationDescription automatically generated with medium confidence - varenicline 11

This is a medication containing varenicline tartrate in spray form. The dosage should be followed as per the package insert. It should be stored at room temperature between 20°C to 25°C (68°Fto77°F) and should not be frozen. The bottle should be discarded after 30 days of first use. It is meant for intranasal use only. The name of the manufacturer is Oyster Paint Pharma Inc.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.