Product Images Upneeq
View Photos of Packaging, Labels & Appearance
- Oxymetazoline Structural Formula - upneeq 1
- Figure G - upneeq 10
- UPNEEQ 30 ct Carton - upneeq 11
- UPNEEQ Trade Foil Pouch - upneeq 12
- upneeq 13
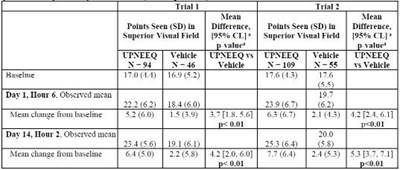
- Table - upneeq 2
- Oxymetazoline Image - upneeq 3
- Figure A - upneeq 4
- Figure B - upneeq 5
- Figure C - upneeq 6
- Figure D - upneeq 7
- Figure E - upneeq 8
- Figure F - upneeq 9
Product Label Images
The following 13 images provide visual information about the product associated with Upneeq NDC 73687-062 by Rvl Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
UPNEEQ Trade Foil Pouch - upneeq 12

UPNEEQ is a prescription ophthalmic solution containing 0.1% oxymetazoline hydrochloride for topical use in the eye. The usual dosage is one drop per day, to be discarded immediately after dosing, and the single-use container should be stored between 20°C-25°C. UPNEEQ is sterile and non-preserved, and included in the package is a single 0.3 mL container. The product is manufactured by Pharmaceuticals, Inc. located in Bridgewater, NJ.*
upneeq 13
UPNEEQ is a medication containing loymetazoine fydochloride. There is additional text that appears to be a percentage but is not readable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.