Product Images Vardenafil Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 19 images provide visual information about the product associated with Vardenafil Hydrochloride NDC 73750-104 by Qyk Brands Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
QYK VARDENAFIL HYDROCHLORIDE 102 10

This is a description of Vardenafil Hydrochloride Tablets, which has a strength of 2.5 mg, and is distributed by QYK Brands. It is manufactured by Macleods Pharmaceuticals Ltd. in Daman, India. The usual dosage is one tablet per day, and more information on dosage and administration can be found in the Prescribing Information. The tablets should be stored at 20-25°C, with excursions permitted to 15-30°C. This appears to be a leaflet that will be distributed to patients, and it contains some basic information on the medication.*
QYK VARDENAFIL HYDROCHLORIDE 102 30

This is a description of a medication called Vardenafil Hydrochloride Tablets, which come in 2.5 mg film-coated tablets. This medication is intended for use as needed but not more than once per day. Patients should refer to the Prescribing Information for Dosage and Administration. The tablets should be stored at a temperature between 20°C to 25°C and can be kept at 15°C to 30°C. QYK Brands distributes these tablets, which are manufactured by Macleods Pharmaceuticals Ltd. in Daman, India.*
QYK VARDENAFIL HYDROCHLORIDE 103 10

This is a description of a medication called Vardenafil Hydrochloride Tablets, with each tablet containing 5mg of Vardenafil, distributed by QYK Brands from Garden Grove, California. The suggested dosage is one tablet as needed, no more than once per day. Further information on dosage and administration can be found in the Prescribing Information. The medication should be stored at 20-25°C (68-77°F) with excursions allowed between 15-30°C (59-86°F). Macleods Pharmaceuticals Ltd. in Daman, India is identified as the manufacturer. This text is a leaflet to be dispensed to each patient.*
QYK VARDENAFIL HYDROCHLORIDE 103 30

This is a description of Vardenafil Hydrochloride Tablets with a dosage of 5 mg per film-coated tablet. The usual dosage is one tablet per day as needed. The leaflet contains prescribing information and dispensing instructions for each patient. The tablets are manufactured by Macleods Pharmaceuticals Ltd. in India and distributed by QYK Brands in Garden Grove, CA. The correct storage temperature is between 20° to 25°C, with permitted excursions to 15" to 30°C, according to USP Controlled Room Temperature guidelines.*
QYK VARDENAFIL HYDROCHLORIDE 104 10

This is a description of a medication called Vardenafil Hydrochloride Tablets, which are contained in a bottle with 100 tablets. Each film-coated tablet contains 10 mg of Vardenafil. Patients are instructed to take one tablet as needed but no more than once per day. The prescribing information should be consulted for dosage and administration instructions. The medication must be stored between 20° to 25°C but may be exposed to excursions between 15" to 30°C. The medication is distributed by QYK Brands in Garden Grove, CA and manufactured by Macleods Pharmaceuticals Ltd. in Daman, India.*
QYK VARDENAFIL HYDROCHLORIDE 104 11

This is a product description for QYK Vardenafil Hydrochloride tablets, each containing 10mg of vardenafil hydrochloride. The usual dosage recommendation is one tablet per day as needed. This product is manufactured by Macleods Pharmaceuticals Ltd. in India and distributed by QYK Brands of Garden Grove, CA. Storage temperatures should be between 20°C to 25°C (68" to 77°F), but excursions to 15°C to 30°C (59" to 86°F) are permitted. Each package of QYK Vardenafil Hydrochloride tablets contains 10 blister strips of 10 tablets, with a barcode on each strip. Further prescribing information can be found with the product.*
QYK VARDENAFIL HYDROCHLORIDE 104 30

This is the patient leaflet for QYK-branded Vardenafil Hydrochloride Tablets. Each tablet contains 10 mg of Vardenafil, to be taken as needed and no more than once per day. It is recommended to refer to the Prescribing Information for Dosage and Administration. The tablets should be stored in 20° to 25°C (68° to 77°F) with permitted excursions to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. The product is distributed by QYK Brands based in Garden Grove, CA, and manufactured by Macleods Pharmaceuticals Ltd. in Daman, India.*
QYK VARDENAFIL HYDROCHLORIDE 105 10

This is a leaflet accompanying 100 film-coated tablets of Vardenafil Hydrochloride, with each tablet containing 20 mg of Vardenafil. It provides information for patients about taking one tablet daily and refers them to additional prescribing information. It also indicates that it should be stored at a controlled temperature between 20° and 25°C and was distributed by QYK Brands, based in Garden Grove, CA, and manufactured by Macleods Pharmaceuticals Ltd. in Daman, India.*
QYK VARDENAFIL HYDROCHLORIDE 105 11

This is a description of a medication called Vardenafil Hydrochloride Tablets. It comes in a bottle of 100 tablets and contains 20mg of Vardenafil per tablet. It is recommended to take one tablet as needed, and no more than once per day. The Tablets should be stored at a temperature between 20°C to 25°C. It's manufactured by Macleods Pharmaceuticals Ltd. in Daman, India and distributed by QYK Brands in Garden Grove, CA.*
QYK VARDENAFIL HYDROCHLORIDE 105 30

This is a barcode label for a medication called "Vardenafil hydrochloride tablets", produced by Macleods Pharmaceuticals Ltd. and distributed by QYK Brands. The label contains dosage and administration instructions, as well as storage guidelines. The barcode indicates the National Drug Code (NDC) number, which is 73750-105-30. It also includes the name of the medication and the name of the pharmaceutical company. A leaflet containing patient information is provided.*
vardenafil-fig-5 - vardenafil fig 5
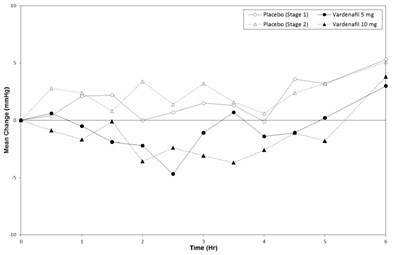
The text does not provide enough information for a description. It appears to be a table or chart related to blood pressure and medication, but the data and context are missing.*
vardenafil-fig-6 - vardenafil fig 6
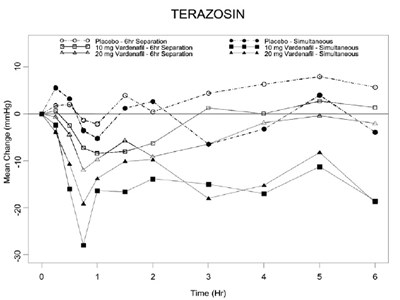
This appears to be a table showing the mean change (in millimeters of mercury) for the drug Terazosin at different times and dosages (90mg, 5mg, and 2-8mg) compared to a placebo. The table also mentions "Verdanafil" and "Separation" but it is not clear how they are related to the data shown.*
vardenafil-fig-7 - vardenafil fig 7
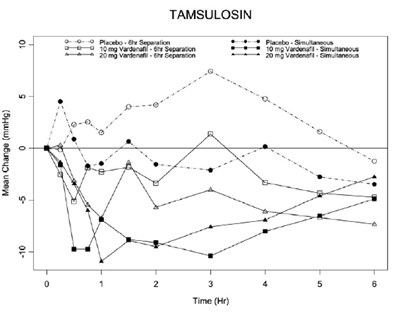
This appears to be a table/statistical data and doesn't provide enough context or information to generate a useful description.*
vardenafil-fig2 - vardenafil fig2

This appears to be a chart or graph displaying the mean change in blood pressure in mmHg over time (in hours) for two groups: one group given a placebo treatment with two different formulations (Simutansos and B Hr Sepamilor) and the other group given a 5 mg dose of the drug Vardenall (Skmuaneous and 8 Hr Separatiar). The graph goes up to 5 mmHg on the positive side and down to -15 mmHg on the negative side. There are tick marks on the X-axis indicating time in hours.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




