Product Images Bylvay
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Bylvay NDC 74528-020 by Albireo Pharma, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - bylvay 02

This is a table that seems to be related to the evaluation of scratching score. It includes different scores from 40 to 0 and two different dosages (40mcg/kg/day and 120mcg/kg/day) of a drug called Odevixibat and a placebo. The table also seems to be organized on a monthly basis.*
PRINCIPAL DISPLAY PANEL - 200 mcg Capsule Bottle Carton - bylvay 07
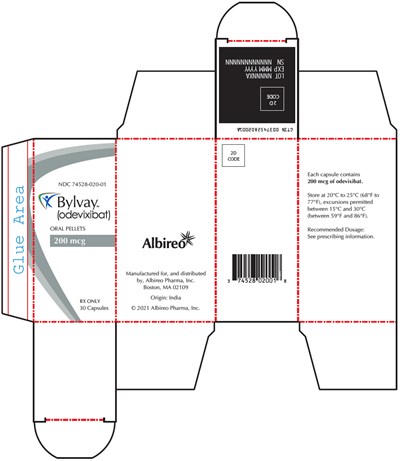
This is a medication description for odevixibat capsules. Each capsule contains 200mg of the medication. The recommended dosage and prescribing information are not available. The capsules are manufactured for and distributed by Albireo Pharma in Boston, MA. The medication is supposed to be stored at a temperature between 20°C to 25°C, with excursions permitted between 15°C and 30°C. The product has a National Drug Code (NDC) of 74528-020-01.*
PRINCIPAL DISPLAY PANEL - 400 mcg Capsule Bottle Carton - bylvay 08

This is a product description for "Bylvay" capsules with the active ingredient "odevixibat". Each capsule contains 400mcg of odevixibat and is intended for prescription use only. The product is manufactured and distributed by Albireo Pharma, Inc. and the recommended dosage is available in the prescribing information. The product origin is India and it must be stored at a temperature range of 20°C to 25°C (68°F to 77°F), with excursions permitted between 15°C and 30°C (between 59°F and 86°F).*
PRINCIPAL DISPLAY PANEL - 600 mcg Capsule Bottle Carton - bylvay 09

This is the label of Y Bylvay oral pellets. Each capsule contains 600mg of odevixibat. The recommended dosage is indicated in the prescribing information. The medication should be stored at a temperature range of 20°C-25°C (68°F-77°F), with excursions between 15°C-30°C (59°F-86°F) permitted. The manufacturer and distributor of the medication is Albireo Pharma, Inc. The NDC number is 74528-060-01.*
PRINCIPAL DISPLAY PANEL - 1,200 mcg Capsule Bottle Carton - bylvay 10
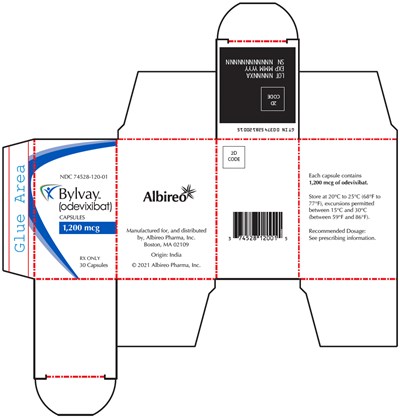
This is a description of Bylvay capsules manufactured by Albireo Pharma. It contains 30 capsules of odevixibat with a recommended dosage that should be obtained from the prescribing information. The NDC number is 74528-120-01, and each capsule contains 1,200 meg of odevixibat. The capsules should be stored between 20°C to 25°C (68°F to 77°F), or between 15°C and 30°C (between 59°F and 86°F) with permissible excursions.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.