FDA Label for Sanitizing Wipes
View Indications, Usage & Precautions
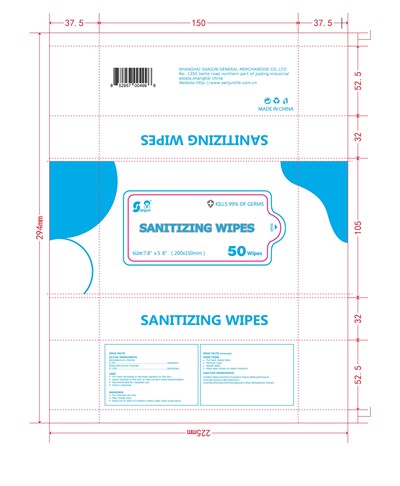
Sanitizing Wipes Product Label
The following document was submitted to the FDA by the labeler of this product Shanghai Sanjun General Merchandise Co., Ltd.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient
Benzalkonium Chloride 0. 1 %
Didecyldimonium Chloride 0.1 5%
Otc - Purpose
Antiseptic
Surfactant
Use
●For hand sanitizing to decrease bacteria on the skin.
●Apply topically to the skin to help prevent cross contamination.
●Recommended for repeated use.
Dries in seconds.
Warning
●For extemal use only
●May iritate eyes
Otc - Keep Out Of Reach Of Children
Keep out of reach of children unless under
adult supervision
Directions
●Pull back reseal label.
●Remove wipe.
。Reseal label.
●Keep label closed ro retain moisture.
Inactive Ingredients
Purified Water, Alcochol,Propylene Glycol,Didecyldimonium
Chloride,Glycerin, Benzalkonium
Chloride,Bronopol,Ethylhexylglycerin,Aloe Barbadensis Extract
Package Label.Principal Display Panel
* Please review the disclaimer below.