Product Images Balcoltra
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Balcoltra NDC 75854-602 by Avion Pharmaceuticals, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Molecular Structures - image 01
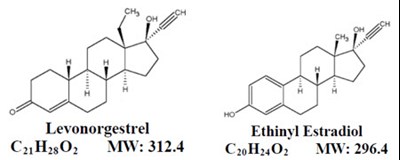
Levonorgestrel Ethinyl Estradiol are two types of female hormones that are used in combination as a form of birth control. The first chemical, Levonorgestrel, belongs to a class of drugs known as progestins and the second chemical, Ethinyl Estradiol, belongs to a class of drugs known as estrogens. These hormones prevent ovulation and make it difficult for sperm to reach the egg, therefore reducing the chances of pregnancy. The molecular formula and weight of Levonorgestrel is CyuHy0 with a weight of 3124, while the molecular formula and weight of Ethinyl Estradiol is CyuHnO with a weight of 296.4.*
Instructions for Use - Figure 2 - image 05
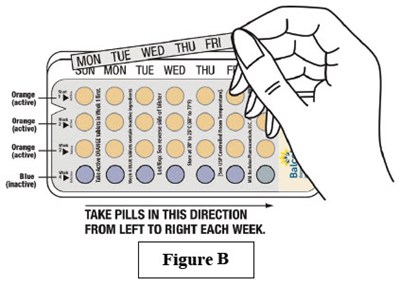
The text provides a direction on how to take pills, “FROM LEFT TO RIGHT EACH WEEK” and mentions “Figure B”. It is likely a part of a medication package insert or prescription label.*
Principal Display Panel - Carton Front - image 06

S Balcoltra is a 28-day regimen birth control pill that prevents pregnancy. It contains levonorgestrel and ethinyl estradiol tablets, USP, and ferrous bisglycinate tablets. It does not protect against HIV or other sexually transmitted diseases. The product is available in 1 blister pack with 28 tablets, and its NDC is 75854-602-28. Women who are over 35 years old and smoke have a higher risk of serious cardiovascular events from the combination of oral contraceptives.*
Principal Display Panel - Carton Back - image 07

This is a description of a medication called Balcoltra contained in a blister pack with 28 tablets to be used in a 28-day regimen. Each pack contains a combination of hormones and instructions for use, which should be included with each dispenser. The medication comprises 21 orange tablets containing vonorgestrel and ethinylestradiol and 7 blue placebo tablets containing ferrous bisglycinate. The medication is to be used once daily for two consecutive days, followed by one blue placebo tablet for seven consecutive days. The medication's usual dose is indicated, and the prescribing information is provided. It should be kept out of reach of children, and in case of accidental overdose, a physician or poison control center should be contacted. The manufacturer is AbnPamceuicals LLC in Alpharetta, GA, and Novut Laboataies in Nantong, China.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


