Product Images Procysbi
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Procysbi NDC 75987-100 by Horizon Therapeutics Usa, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 25 mg Capsule Bottle Label - procysbi 02

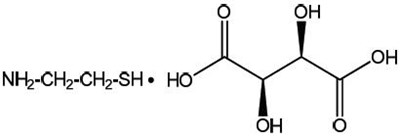
Each capsule of PROCYSBI contains 74mg of cysteamine bitartrate equivalent to 25mg cysteamine. These delayed-release capsules must be refrigerated and dispensed only in original packaging. Keep out of reach of children and store at a temperature of 2°C to 8°C (35°F to 457). PROCYSBI is distributed by PRSI eye. Attention patients, discard unused portion after 4 months and store at room temperature between 20°C to 25°C (B8°F to 77°F) and protect from light and moisture. The prescription information should be sought before taking PROCYSBI. In case of doubt, consult a pharmacist.*
PRINCIPAL DISPLAY PANEL - 75 mg Capsule Bottle Label - procysbi 03

This text is a description of a medicine called PROCYSBI, which is contained in capsules. The capsules contain 221mg of cysteamine and must only be dispensed in o)) bitartrate equivalent to 75mg of cysteamine. The medicine must be refrigerated at 2°C to 8°C (36°F to 46°F) before dispensing and has a 4-month expiration date. Patients should store the medicine at room temperature between 20°C to 25°C (68°F to 77°F) and avoid exposure to light and moisture. Unused portions of the medicine should be discarded 4 months after the dispensing day. This medicine is distributed by Horizon Therapeutics USA, Inc. from Italy.*
procysbi 04

This is a drug called PROCYSBI, which contains cysteamine bitartrate, and is dispensed as delayed-release oral granules. The packet contains 221 mg of cysteamine bitartrate, equivalent to 75 mg of cysteamine. It is advisable to store the drug at room temperature and keep out of reach of children. The National Drug Code (NDC) for this medication is 75987-140-13. The drug is distributed by Horizon Therapeutics USA, Inc. in Deerfield, IL60015. It is important to dispense only in the original packaging.*
procysbi 05

This is a description of a medication called PROCYSBI which is a delayed-release oral granule formulation containing cysteamine bitartrate. It is dispensed in original packaging and each packet contains 884mg or 300g of the medication. It should be kept at a temperature between 20°C to 25°C. Unused packets should be discarded after the expiration date. The medication should be kept out of reach of children. The manufacturer is located in Deerfield, IL. The National Drug Code (NDC) for this medication is 75987-145-13.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.