Product Images Midazolam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 19 images provide visual information about the product associated with Midazolam NDC 76045-001 by Fresenius Kabi Usa, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
mid08 0005 02

Midazolam Open-ring Form is a form of the medication midazolam that is used for sedation and anesthesia in medical procedures. This form of midazolam has an "open-ring" structure, meaning that it is in a more unstable and reactive state compared to other forms of the drug. When administered, midazolam open-ring form quickly converts to the more stable and active form of midazolam, which allows for rapid onset of sedation.*
mid08 0005 07

This text indicates the location information of Fresenius Kabi in Lake Zurich, IL 60047.*
mid08 0005 08

This is a product description for Rxonly NDC 76045-211-20, a medication that is administered through intravenous or intramuscular use only. The medication is delivered in 24 single-dose prefilled syringes, with a 2 mL capacity. The package includes a caution to not place the syringe on a sterile field and all unused portions of the medication should be discarded. There are some additional characters that appear to be garbled or not in English, so the description may not be complete.*
mid08 0005 09

This is the package insert for a single-dose pre-filled syringe from the company Fresenius Kabi. The syringe is intended for intravenous or intramuscular use only and should be stored at room temperature and protected from light. The dosage should be determined according to the package insert and any unused portion should be discarded. There is an expiration date provided, but it is not visible in the text.*
mid08 0005 11

This appears to be a medication with the National Drug Code (NDC) 76045-001-20 that is meant for intravenous or intramuscular use. The medication is available in a package containing 24 single-dose prefilled syringes that should not be placed on a sterile field. The text includes some distorted characters and some numerical values that appear to be part of a code or lot number. Without further context, it is not possible to determine the specific medication or its purpose.*
mid08 0005 12

This text contains instructions and precautions for the use of a medical product, WoC 76045-001.00. It is a single-dose prefilled syringe designed for intravenous or intramuscular use only. The product should be stored between 20° to 25°C, protected from light. The typical dosage requirements are mentioned in the package insert. The syringe should not be placed on a sterile field, and any unused portion should be discarded. The text ends with an expiration date code and an RN number.*
mid08 0005 13

This is a pharmaceutical product manufactured by Fresenius Kabi. It comes in a 2mL single-dose and is intended for intravenous or intramuscular use only. The product is only available with a prescription, denoted as "Rx only". The other characters are likely product codes or identification numbers.*
mid08 0005 14

This is a description of Midazolam Injection, USP with NDC code 76045-002-10. It is a prescription medicine that can only be used for intravenous or intramuscular administration. The medication is available in 24 x 1 mL single-dose prefilled syringes. The text includes instructions to discard the unused portion and not to place the syringe on a sterile field.*
mid08 0005 15
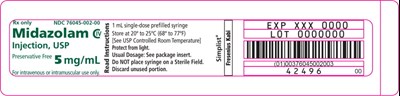
Simplist midazolam injection is a sterile solution of midazolam for intravenous or intramuscular administration. The package includes a disposable, single-dose prefilled syringe that contains 1 mg/mL midazolam. It is a controlled room temperature drug and should be stored between 20°C-25°C (68°F-77°F). The unused portion of the syringe should be discarded. The preservative-free drug should be protected from light. For dosage information, refer to the package insert. This product is manufactured by Fresenius Kabi. The expiry date and lot number are mentioned.*
mid08 0005 16

This is a description of a pharmaceutical product. It is a single-dose injection for intravenous or intramuscular use only. The product contains 5 mg/mL of midazolam and is preservative-free. The packaging comes in two options: 1 mL or 0.5 mL. The manufacturer is Fresenius Kabi. The rest of the text is not relevant to the product description.*
mid08 0005 17

This appears to be a description of a medication with the NDC code 76045-003-20. It is a preservative-free injection of 10nm/2m (5mg/mL) midazolam for intravenous or intramuscular use only. The medication is labeled "No Coat Area". Any unused portion should be discarded. The remaining text is not readable.*
mid08 0005 18

This appears to be a description of a medication called Midazolam. It is an injection that comes in a single-dose pre-filled syringe with a dosage of 10 mg/2 mL. The medication should be stored at a temperature range of 20°-25°C and protected from light. The prescription may be administered intravenously or intramuscularly and is preservative-free. The package insert should be consulted for the appropriate dosage. Any unused portions of the medication should be discarded, and the instructions should be read carefully.*
mid08 0005 19

This is a description of a 2mL single-dose of Midazolam, meant for intravenous or intramuscular use only, with a prescription requirement. The medication is manufactured by Fresenius Kabi and contains 10mg/mL of Midazolam. The product is preservative-free, and the label indicates the lot number as 403561.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





