Product Images Haloperidol
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Haloperidol NDC 76045-737 by Fresenius Kabi Usa, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
hal07 0005 02
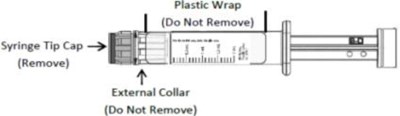
The text describes the labels on some objects. The first label reads "Plastic Wrap (Do Not Remove)" and the second is a directional instruction to remove the tip cap of a syringe. The third label reads "External Collar (Do Not Remove)".*
hal07 0005 06

Description: This appears to be the text from the packaging of a medical product, identified by the NDC number "Rxony NDC7604573701g". The product is a single-dose syringe with a volume of 1L, intended for intramuscular use. The text includes instructions to store the syringe at a temperature between 20°C and 25°C and to not freeze it. There is a lot number provided, as well as an expiration date represented by "EXP_XXX 0000". The text also mentions a package insert that provides information on the usual dosage.*
hal07 0005 07
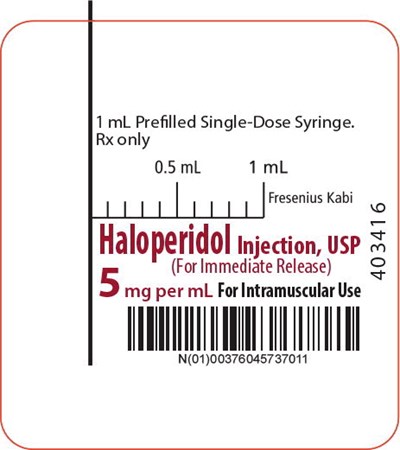
The text describes a single-dose syringe manufactured by Fresenius Kabi with 5 mg per mL of Haloperidol injection, usp intended for intramuscular use. The syringe size is 1 mL and is only for prescription (Rx only). The syringe is also available in 0.5 mL to 1 mL doses. The provided number, N(01)00376045737011, is unclear what it represents.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.