Product Images Kyprolis
View Photos of Packaging, Labels & Appearance
- 3. Use a 21 gauge or larger gauge needle (0.8 mm or smaller external diameter needle) to aseptically reconstitute each vial by slowly injecting 29 mL (for 60 mg vial) or 15 mL (for 30 mg vial) Sterile Water for Injection, USP, through the stopper and directing the solution onto the INSIDE WALL OF THE VIAL to minimize foaming. - kyprolis 01
- The following structural formula for Carfilzomib is a modified tetrapeptidyl epoxide, isolated as the crystalline free base. The chemical name for carfilzomib is (2S)-N-((S)-1-((S)-4-methyl-1-((R)-2-methyloxiran-2 yl)-1-oxopentan-2-ylcarbamoyl)-2-phenylethyl)-2-((S)-2-(2-morpholinoacetamido)-4 phenylbutanamido)-4-methylpentanamide. - kyprolis 02
- Figure 1: Kaplan-Meier Curve of Progression-Free Survival in Study 1 - kyprolis 03
- Figure 2: Kaplan-Meier Curve of Interim Overall Survival in Study 1 - kyprolis 04
- Figure 3: Kaplan-Meier Plot of Progression-Free Survival in Study 2 - kyprolis 05
- Figure 4: Kaplan-Meier Plot of Overall Survival in Study 2 - kyprolis 06
- kyprolis 07
- PRINCIPAL DISPLAY PANEL1 Single-Dose VialNDC 76075-102-01AMGEN®Kyprolis®(carfilzomib) for Injection30 mg/vial30 mg/vialSingle-dose vial.Discard unused portion.For Intravenous Administration OnlyRx Only - kyprolis 08
- PRINCIPAL DISPLAY PANEL1 Single-Use VialNDC 76075-101-01AMGEN®Kyprolis®(carfilzomib) for Injection60 mg/vial60 mg/vialSingle-use vial.Discard unused portion.For Intravenous Administration OnlyRx Only - kyprolis 09
- kyprolis 10
- kyprolis 11
- kyprolis 12
Product Label Images
The following 12 images provide visual information about the product associated with Kyprolis NDC 76075-103 by Onyx Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
3. Use a 21 gauge or larger gauge needle (0.8 mm or smaller external diameter needle) to aseptically reconstitute each vial by slowly injecting 29 mL (for 60 mg vial) or 15 mL (for 30 mg vial) Sterile Water for Injection, USP, through the stopper and directing the solution onto the INSIDE WALL OF THE VIAL to minimize foaming. - kyprolis 01
The following structural formula for Carfilzomib is a modified tetrapeptidyl epoxide, isolated as the crystalline free base. The chemical name for carfilzomib is (2S)-N-((S)-1-((S)-4-methyl-1-((R)-2-methyloxiran-2 yl)-1-oxopentan-2-ylcarbamoyl)-2-phenylethyl)-2-((S)-2-(2-morpholinoacetamido)-4 phenylbutanamido)-4-methylpentanamide. - kyprolis 02
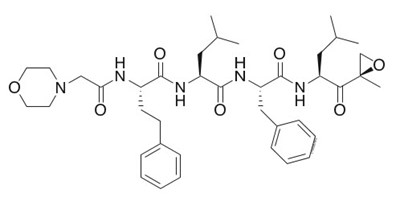
Figure 1: Kaplan-Meier Curve of Progression-Free Survival in Study 1 - kyprolis 03
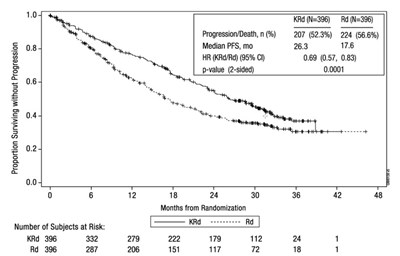
This is a clinical trial report that compares the treatment of two groups of patients (KRd and Rd) with multiple myeloma in terms of progression-free survival (PFS) and mortality. The KRd group had a higher overall PFS and lower mortality rate than the Rd group. The report includes relevant statistics, such as hazard ratio and p-value, as well as a graph displaying the number of subjects at different points in time.*
Figure 3: Kaplan-Meier Plot of Progression-Free Survival in Study 2 - kyprolis 05

This appears to be a table showing the proportion of patients surviving without progression in two different treatment groups ("Kd" and "Vd") with their respective sample sizes and percentages of patients experiencing progression or death. It also shows the median progression-free survival time (in months) and other statistical measures such as hazard ratio and p-value. The text is not clear enough to make sense of the remaining information.*
Figure 4: Kaplan-Meier Plot of Overall Survival in Study 2 - kyprolis 06
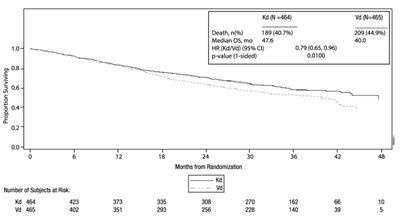
This is a report on the results of a study on Kd and Vd involving 464 and 465 test subjects respectively. The report provides data on the prevalance of death (40.7%), median Os (476), HR (Kd/Vd) and the corresponding 95% confidence interval (0.79 [0.65,0.96]), and the p-value (0.01 - one-sided). The report also includes a graph showing the number of subjects at risk over time. No additional context or content is available for this text.*
kyprolis 07

The text describes the results of a medical study comparing the effectiveness of two dosages of a drug called Kd, administered either twice-weekly at a lower dosage or once-weekly at a higher dosage. The study involved 238 subjects and measured the proportion of patients free from disease progression after different periods of time, among other metrics. The results suggest that the once-weekly dosage was more effective in delaying disease progression, with a lower proportion of patients experiencing progression or death, and a longer median progression-free survival time, compared to the twice-weekly dosage. However, the text does not provide information on the specific medical condition or context of the study.*
PRINCIPAL DISPLAY PANEL1 Single-Dose VialNDC 76075-102-01AMGEN®Kyprolis®(carfilzomib) for Injection30 mg/vial30 mg/vialSingle-dose vial.Discard unused portion.For Intravenous Administration OnlyRx Only - kyprolis 08
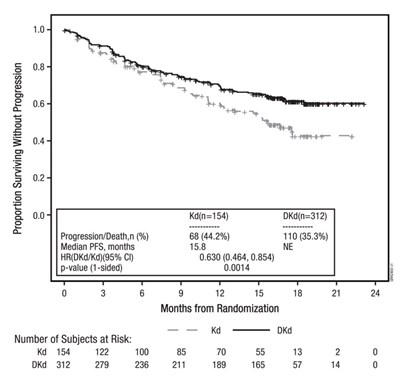
This is a table showing the proportion surviving without progression, progression/death rate, median PFS, HR (DK/Kd), p-value, and the number of subjects at risk for a certain study. The study was conducted for a certain duration, and the number of subjects at risk is provided at different months from randomization. Further information on the study is not available.*
PRINCIPAL DISPLAY PANEL1 Single-Use VialNDC 76075-101-01AMGEN®Kyprolis®(carfilzomib) for Injection60 mg/vial60 mg/vialSingle-use vial.Discard unused portion.For Intravenous Administration OnlyRx Only - kyprolis 09
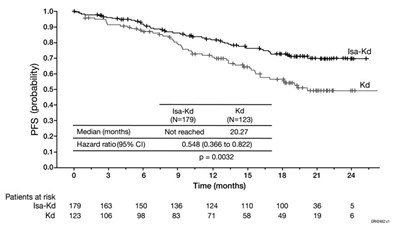
This appears to be a survival analysis of two groups, Isa-Kd and Kd, with a median follow-up time of more than 20 months. The hazard ratio suggests a statistically significant lower risk of an event (death, relapse, etc) in the Isa-Kd group compared to the Kd group. However, without further information on the study design, types of events, and patient characteristics, it is difficult to fully understand the clinical implications of the findings.*
kyprolis 10

Amgen's Kyprolis comes in a single-dose vial containing 10mg of the medication. The dosage is meant for intravenous use only, and the vial should be discarded after use. The drug should be reconstituted with 3.4mL of sterile water for injection USP. Additional information about the drug's use is not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


