Product Images Gammaked
View Photos of Packaging, Labels & Appearance
- Figure 7 - figure7
- Figure 8 - figure8
- 100 mL Carton - gammakedcarton
- Administration - pi admin2
- Step 11 - step11bw
- Step 1 - step1bw
- Step 2 - step2bw
- Step 3 - step3bw
- Step 4 - step4bw
- Step 5 - step5bw
- Step 6 - step6bw
- Step 7 - step7bw
- Step 8 - step8bw
- Step 9a - step9abw
- Step 9b - step9bbw
- 100 ml Vial - vial
Product Label Images
The following 16 images provide visual information about the product associated with Gammaked NDC 76125-900 by Kedrion Biopharma, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 7 - figure7
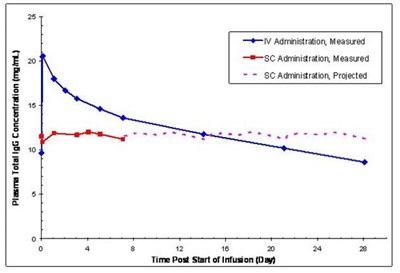
This is a graph that shows the concentration of plasma total IgG in mg/mL over time following different modes of administration (IV, SC) of an unknown substance. There is also a projected line for SC administration. However, without additional context, it is impossible to determine the exact substance being administered or the purpose of the study.*
Figure 8 - figure8
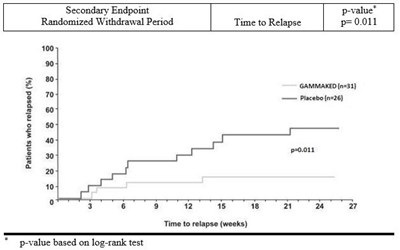
This is a graph showing the time to relapse during a randomized withdrawal period for patients taking GAMMAKED (n=31) compared to placebo (n=26). The secondary endpoint p-value for time to relapse is 0.011, based on a log-rank test. The x-axis shows time in weeks and the y-axis shows the percentage of patients who relapsed. There is also a table next to the graph displaying the number and percentage of patients who relapsed at various time points.*
100 mL Carton - gammakedcarton

This is a description of a medication called Gammaked, an immune globulin injection used for intravenous or subcutaneous administration. It comes in a package that includes a single-dose vial with a dosage and administration guide. The product has no preservatives, has 98% immunoglobulin, and around 15 mg of glycoprotein. The medication should be discarded if there is tampering or if the seal is absent. The manufacturer is Grifols Therapeutics LLC. The medication should be stored between 2-8°C and not exceed 25°C. If stored properly, the product has a shelf life of 36 months.*
100 ml Vial - vial

Gammaked is an injection solution for infusion, used for intravenous or subcutaneous administration, with dosage and administration information included in the packaging. Storage at temperatures between 2-8°C for up to 24 months is recommended. The product must be used immediately upon opening. Lot and batch number information is provided on the packaging. Patient and physician discussions about the risks and benefits of the product are recommended.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.











