Product Images Levetiracetam
View Photos of Packaging, Labels & Appearance
- str - levetiracetam tablets 1
- Levetiracetam 750 mg Tablets, USP - levetiracetam tablets 10
- fig 1 - levetiracetam tablets 2
- fig 2 - levetiracetam tablets 3
- fig 3 - levetiracetam tablets 4
- fig 4 - levetiracetam tablets 5
- fig 6 - levetiracetam tablets 7
- Levetiracetam 250 mg Tablets, USP - levetiracetam tablets 8
- Levetiracetam 500 mg Tablets, USP - levetiracetam tablets 9
Product Label Images
The following 9 images provide visual information about the product associated with Levetiracetam NDC 76282-248 by Exelan Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Levetiracetam 750 mg Tablets, USP - levetiracetam tablets 10

Levetiracetam Tablets, USP is a medication used to treat seizures. The usual dosage and storage instructions are included on the packaging insert. The medication should be dispensed in a child-resistant container and stored at 25°C (77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature]. The medication is manufactured for Exelan Pharmaceuticals, Inc. in Lawrenceville, GA 30046 and manufactured by InvaGen Pharmaceuticals, Inc. in Hauppauge, NY 11788.*
fig 1 - levetiracetam tablets 2
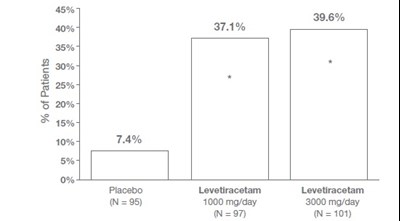
This appears to be a chart showing various percentages of patients, as well as percentages attributed to specific treatments (placebo, Levetiracetam at 1000mg/day, and Levetiracetam at 3000mg/day). It is unclear what the context or purpose of the chart is, as no further information is provided.*
fig 2 - levetiracetam tablets 3
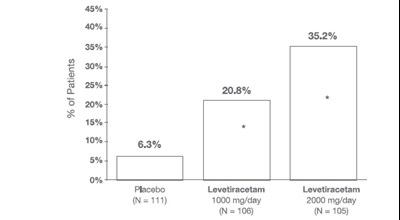
This appears to be a chart or graph showing the percentages of patients in different treatment groups. The treatments are likely related to seizures or epilepsy since one of the groups is labeled with a medication name, Levetiracetam. The chart also shows the number of patients in each group.*
fig 4 - levetiracetam tablets 5
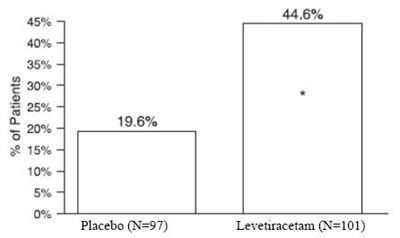
This is a table showing percentages of patients in a medical study. It compares placebo to a drug called Levetiracetam. The percentages given are 45%, 40%, 35%, 30%, 25%, and 20% for patients in both groups. There are also symbols for percent, at, and a registered trademark. In addition, there are specific percentages of 10% and 19.6% mentioned, but no context is given.*
fig 6 - levetiracetam tablets 7
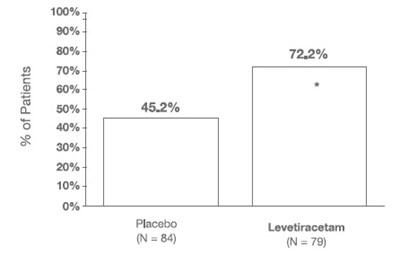
This is a chart or table depicting the percentage of patients, indicated by numerical values, receiving either Placebo or Levetiracetam. The total number of patients included in the study appears to be 84. Further information or context is not available with only these details.*
Levetiracetam 250 mg Tablets, USP - levetiracetam tablets 8
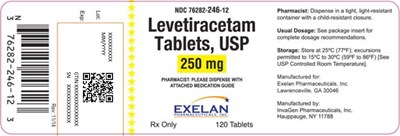
The text contains a medication guide for Levetiracetam tablets, USP 250mg. The medication should be dispensed in a light-resistant container with a child-resistant closure by the pharmacist. The recommended storage temperature is at 25°C (77°F) with limited excursions permitted. The tablets are manufactured by InvaGen Pharmaceuticals, Inc. and distributed by Exelan Pharmaceuticals, Inc. in Lawrenceville, GA.*
Levetiracetam 500 mg Tablets, USP - levetiracetam tablets 9

Levetiracetam Tablets, USP is a prescription medication manufactured by InvaGen Pharmaceuticals, Inc. and distributed by Exelan Pharmaceuticals, Inc. The medication is enclosed in a container with a child-resistant closure and an attached medication guide. The package contains 120 tablets and must be stored at a controlled temperature between 15°C to 30°C (59°F to 86°F). Usual dosage instructions can be found in the package insert. Pharmacists are advised to dispense the medication in a tight and light-resistant container.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

