Product Images Topiramate
View Photos of Packaging, Labels & Appearance
- Figure 1: Kaplan-Meier Estimates of Cumulative Rates for Time to First Seizure - topiramate fig1
- Table 12: Efficacy Results in Double-Blind, Placebo-Controlled, Add-On Epilepsy Trials - topiramate fig2
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg (60 Tablets Bottle) - topiramate fig3 inv
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg (60 Tablets Bottle) - topiramate fig4 inv
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg (60 Tablets Bottle) - topiramate fig5 inv
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg (60 Tablets Bottle) - topiramate fig6 inv
- chemical structure - topiramate str
Product Label Images
The following 7 images provide visual information about the product associated with Topiramate NDC 76282-279 by Exelan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Table 12: Efficacy Results in Double-Blind, Placebo-Controlled, Add-On Epilepsy Trials - topiramate fig2
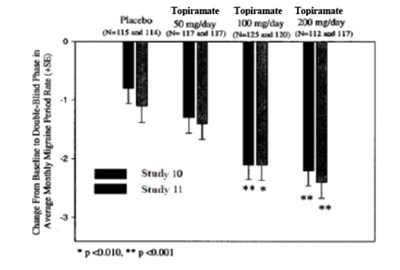
This appears to be a table or chart showing the effectiveness of Topiramate at different dosage levels for a study labeled "Study 10." The effectiveness is likely measured by a percent or score, but that information is not present in the excerpt. The study had a notable amount of participants (115 to 117) at each dosage level, except for one group with only 25 participants. The text, unfortunately, does not provide additional information beyond the medication name, dosages, and study findings.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 25 mg (60 Tablets Bottle) - topiramate fig3 inv

This is a description of a medication called topiramate, specifically the 25mg tablets. The bottle contains 1000 tablets and comes with an attached medication guide. It should be dispensed in a tight container and stored between 20° to 26°C (68° to 77°F). The manufacturer is InvaGen Pharmaceuticals, a subsidiary of Cipla Ltd, located in Hauppauge, NY. The medication is manufactured for Exelan Pharmaceuticals, Inc. based in Boca Raton, FL. For more information on dosage and prescribing, refer to the accompanying product literature.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg (60 Tablets Bottle) - topiramate fig4 inv
This is a description for medication containing topiramate, USP available in tablet form with dosage information and prescribing guidelines found in the accompanying literature. It should be stored in a tight container at a temperature of 20° to 25°C. The medication is manufactured by InvaGen Pharmaceuticals, Inc., a subsidiary of Cipla Ltd. for Exelan Pharmaceuticals, Inc. in Boca Raton, FL. This medication is available with a prescription only and the pack contains 1000 tablets.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg (60 Tablets Bottle) - topiramate fig5 inv

This is a description of "Topiramate Tablets, USP" produced by Exelan Pharmaceuticals, Inc. Each tablet contains Topiramate, USP 100mg, and it is manufactured by InvaGen Pharmaceuticals, Inc. It includes a medication guide and should be stored in a tightly closed container at 20°C to 25°C. The dosage information, along with other prescribing information, is detailed in the accompanying product literature. It is a prescription drug, and a pharmacist's dispensation is required.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg (60 Tablets Bottle) - topiramate fig6 inv
This is a description of a medication called "Topiramate Tablets, USP" produced by Exelan Pharmaceuticals, Inc. The tablets are available in a bottle of 1000 and each tablet contains 200mg of topiramate. This text also includes a NDC code indicating product registration. For safety reasons, the pharmacist is required to dispense the medication with an attached medication guide. There are instructions on how to store the medication available on the label.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

