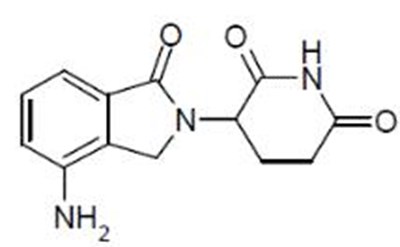
Product Images Lenalidomide
View Photos of Packaging, Labels & Appearance
- Image - 084916cb cd26 4942 bc5b 6d4736594420 01
- 1 - 084916cb cd26 4942 bc5b 6d4736594420 02
- 2 - 084916cb cd26 4942 bc5b 6d4736594420 03
- 3 - 084916cb cd26 4942 bc5b 6d4736594420 04
- 4 - 084916cb cd26 4942 bc5b 6d4736594420 05
- 5 - 084916cb cd26 4942 bc5b 6d4736594420 06
- 6 - 084916cb cd26 4942 bc5b 6d4736594420 07
- image-1 - 084916cb cd26 4942 bc5b 6d4736594420 08
- Container-5mg-genvion - 084916cb cd26 4942 bc5b 6d4736594420 09
- container-10mg-genvion - 084916cb cd26 4942 bc5b 6d4736594420 10
- container-15mg-genvion - 084916cb cd26 4942 bc5b 6d4736594420 11
- container-25mg-genvion - 084916cb cd26 4942 bc5b 6d4736594420 12
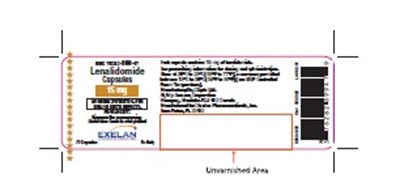
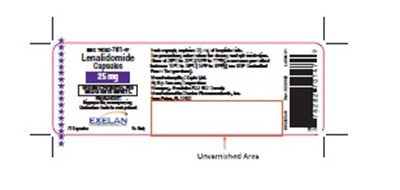
Product Label Images
The following 12 images provide visual information about the product associated with Lenalidomide NDC 76282-701 by Exelan Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
1 - 084916cb cd26 4942 bc5b 6d4736594420 02

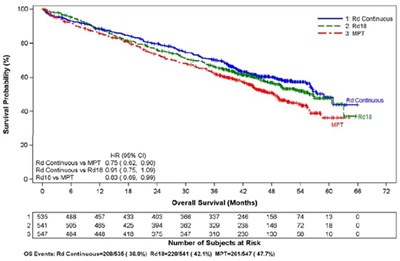
The text is a combination of numbers, symbols, and characters that do not form a coherent and understandable description. The extracted text may be a table consisting of data related to survival probability and progression-free survival of some subjects, but without proper formatting, it is not readable. Therefore, the description is not available.*
3 - 084916cb cd26 4942 bc5b 6d4736594420 04

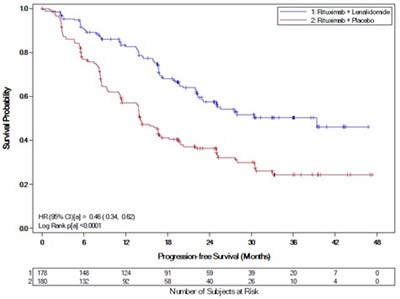
This appears to be a comparative study between Lenalidomide and Placebo done to evaluate the Progression-free Survival (PFS) in subjects. The results show that subjects who received Lenalidomide had a significantly higher PFS compared to Placebo. The HR(95%CI) was 0.38(0.28,0.50) and the median PFS for Lenalidomide was 63.6 months (52.8, not estimated) whereas for Placebo it was 18.5 months (30.0). There were 231 subjects who received Lenalidomide and 229 who received Placebo. The percentage of PFS events was lower in the Lenalidomide group than the placebo group.*
4 - 084916cb cd26 4942 bc5b 6d4736594420 05

This is a clinical trial result comparison between Lenalidomide and placebo. The trial evaluated progression-free survival between the two groups, with Lenalidomide showing a statistically significant improvement. The data includes hazard ratios, median survival times, and the number of subjects at risk.*
5 - 084916cb cd26 4942 bc5b 6d4736594420 06

This text provides information on the proportion of subjects who progressed (HR) when using a treatment with Lenalidomide/Dex. The results suggest that only 25% of subjects progressed with a hazard ratio (HR) of 0.285 and a confidence interval (CI) of 0.210 to 0.386. Additionally, the Log Rank p-value was less than 0.001. The time to progression in months is also given for treatment at levels of 10 and 20.*
6 - 084916cb cd26 4942 bc5b 6d4736594420 07

The text describes a graph showing the proportion of subjects at different time intervals (100%, 75%, 50%, and 25%) and a comparison of the Lenalidomide/Dex treatment and Placebo/Dex treatment for time to progression in months. The HR (hazard ratio) is reported as 0.324 with a 95% confidence interval of 0.240-0.438 and a Log Rank p-value of less than 0.0001.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.