Product Images Benztropine Mesylate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Benztropine Mesylate NDC 76385-104 by Bayshore Pharmaceuticals Llc (nj), such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
1mg 1000tabs - 1mg1000ct

This is a medication for parkinsonism and drug-induced extrapyamidal disorders. The tablets contain Benziopine Mesylte, USP and the recommended dosage is 1-4mg once or twice a day for adults. The medication should be stored at room temperature and kept out of reach of children. Side effects should be reported to the FDA and the medication is only available with a prescription. The medication is manufactured by Bayshore Pharmaceuticals LLC and comes in a container with a child-resistant closure.*
1mg 100tabs - 1mg100ct

This is a medication called Benztropine Mesylate, available in the form of tablets, with a usual adult dosage of 1 to 4 mg per day for drug-induced extrapyramidal disorders. It should be stored at room temperature and kept out of the reach of children. Any side effects should be reported to the doctor or FDA. The pack is containing 100 tablets and manufactured by Bayshore Pharmaceuticals LLC with a National Drug Code (NDC) of 76385-104-01.*
2mg 1000tabs - 2mg1000ct

This is a description for a medication called Benztropine Mesylate Tablets, which provide relief for parkinsonism and drug-induced extrapyramidal disorders among adults. It is recommended to take 1-4mg once or twice a day. The tablets should be stored at controlled room temperature and kept in a child-resistant container. Patients should consult their doctor regarding any side effects and can report them to the FDA. The package contains 1000 tablets and the NDC number is 76385-105-10. The document also includes administrative details such as the revision and location.*
2mg 100tabs - 2mg100ct

This is a description of a medication containing Bendopine Mesylate, indicated for the treatment of induced extrapyramidal disorders and accompanying symptoms. The medication is in tablet form, with each tablet containing 2mg of the active ingredient. The National Drug Code (NDC) for the medication is 76385-105-01. The medication should be stored at room temperature, in a closed container as defined by the USP using Rx only. The packaging contains 100 tablets and patients should keep the medication out of reach of children. Side effects, if any, should be discussed with a doctor and can also be reported to the FDA.*
Chemical structure - chemstructure
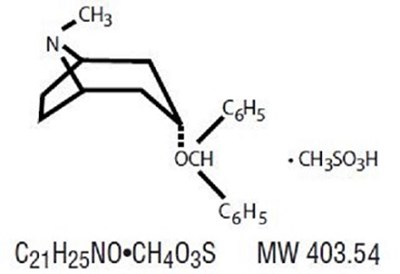
The text is a chemical formula that represents a molecule with the following components: - Nitrogen (N) - Methyl group (CH3) attached to Nitrogen (N) - Oxygen (O) double-bonded to Carbon (C) - Methylene group (CH2) attached to Oxygen (O) and Carbon (C) - Benzene ring (C6H5) attached to Carbon (C) with the Oxygen (O) and Methylene group (CH2) also attached to this Carbon (C) - Nitrogen (N) double-bonded to Carbon (C) that is also attached to three Hydrogen atoms (H) - Carbon (C) attached to two benzene rings (C6H5), with a Nitrogen atom (N) and a Sulphur atom (S) attached to one of the rings. The molecular weight of this compound is 403.54. The name of the molecule is not available.*
0.5mg 1000tabs - point5mg1000ct

This is a medication description for Benztropine Mesylate Tablets, USP. It provides dosage information for treating parkinsonism and drug-induced extrapyamidl disorders. It also includes storage instructions, dispensing details, and precautions. The tablets come in a well-dosed container and must be kept away from children. The side effects can be reported to FDA at 1-800-FDA-1088. The NDC number for this medication is 76385-103-10 and it's available by prescription only.*
0.5mg 100tabs - point5mg100ct

Benztropine Mesylate is a medication commonly used to treat drug-induced extrapyramidal disorders. The usual adult dosage range is 1 to 4 mg twice a day. The medication should be stored at room temperature in a child-resistant container. Individuals are advised to seek medical advice from a physician about any potential side effects. It is essential to keep this medication and all other medications out of the reach of children. The tablets are dispensed in a well-dosed container. An NDC code of 76385-103-01 is included on the packaging, and this medication is only available with a prescription. The label includes contact information, including the phone number for reporting side effects to the FDA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.