Product Images Tadalafil
View Photos of Packaging, Labels & Appearance
- spl-tadalafil-figure-1 - spl tadalafil figure 1
- spl-tadalafil-figure-2 - spl tadalafil figure 2
- spl-tadalafil-figure-3 - spl tadalafil figure 3
- spl-tadalafil-figure-4 - spl tadalafil figure 4
- spl-tadalafil-figure-5 - spl tadalafil figure 5
- spl-tadalafil-figure-6 - spl tadalafil figure 6
- spl-tadalafil-figure-8 - spl tadalafil figure 8
- spl-tadalafil-label-10mg - spl tadalafil label 10mg
- spl-tadalafil-label-20mg - spl tadalafil label 20mg
- spl-tadalafil-label-5mg - spl tadalafil label 5mg
- spl-tadalafil-showbox-1 - spl tadalafil showbox 1
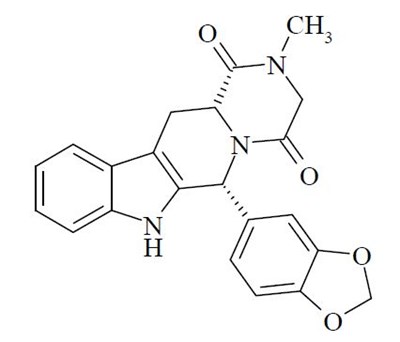
- spl-tadalafil-structure - spl tadalafil structure
Product Label Images
The following 12 images provide visual information about the product associated with Tadalafil NDC 76420-543 by Asclemed Usa, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
spl-tadalafil-figure-3 - spl tadalafil figure 3

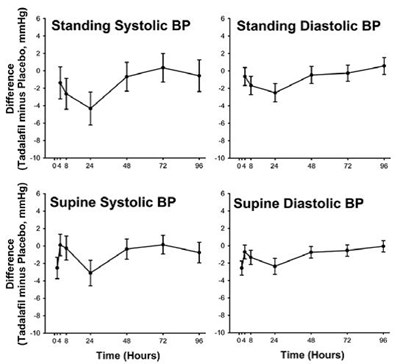
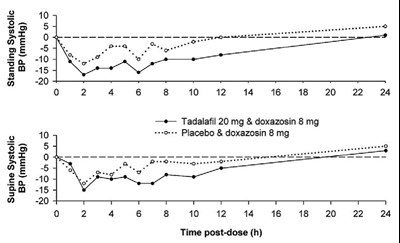
The text provides information on a study involving different doses of a medication called tadalafil and the medication doxazosin. The data includes systolic blood pressure readings (in mmHg) at different time points after administration of the medication combinations, as well as placebos, and specifies the dosages and timing of each. No other context or details are given.*
spl-tadalafil-figure-5 - spl tadalafil figure 5

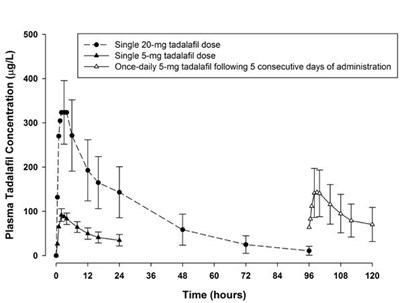
This text provides information on the change in IPSS total score for Tadalafil 5 mg compared to placebo over a certain duration of treatment in weeks. A significant difference was observed, with a p-value of less than 0.01.*
spl-tadalafil-figure-6 - spl tadalafil figure 6
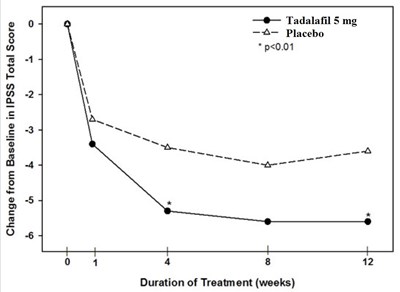
The text describes a graph that shows the change from baseline in IPSS (International Prostate Symptom Score) total score during a 12-week treatment with Tadalafil 5 mg compared to a placebo. The graph shows that there was a statistically significant improvement in IPSS total score with Tadalafil 5 mg compared to placebo (p<0.01).*
spl-tadalafil-figure-8 - spl tadalafil figure 8

The text provides information on the change from baseline in IPSS total score achieved with Tadalafil 5mg vs Placebo. The difference between the two was statistically significant with p<0.01. However, there is no information available about the duration of the treatment.*
spl-tadalafil-label-10mg - spl tadalafil label 10mg

This is a description of Tadalafil Tablets, USP 10mg. The label indicates that the pills were manufactured for Northstar Rx, LLC and distributed by Enovachem from Torrance. The pills are a low sobredlovl shape with a fim-coied abesidepossed numbering of 105 (1T). The package contains 30 tablets. The drug is available only with a prescription and should be kept out of reach of children. The pills should be stored at 20-25C (68-77F) [SEE USP CONTROLLED ROOM TEMP].*
spl-tadalafil-label-20mg - spl tadalafil label 20mg

This label shows information about Tadalafil Tablets, USP 20mg made for Northstar Rx LLC. The NDC number is 76420-544-30, and the quantity is 30 tablets per container. The drug requires a prescription and the package insert should be read before use. The label provides storage instructions and a warning to keep out of reach of children.*
spl-tadalafil-label-5mg - spl tadalafil label 5mg

This is a product description for Tadalafil Tablets, USP 5mg that come in quantities of 30 or 90. The tablets are manufactured by Northstar Rx and have a federal RX SIN drug status, which means they require a prescription to be dispensed. The packaging warns that they are not to be given to children and they should be stored at a controlled room temperature of 20-25C (68-77F). The label includes important information such as the NDC, batch number, and expiration date.*
spl-tadalafil-showbox-1 - spl tadalafil showbox 1

This is a description of a package of Tadalafil Tablets, USP 2.5mg from Northstar Rx, LLC. The package contains 30 tablets and the lot number is 00000000 with an expiration date of 00000000. It is a prescription drug and should not be dispensed without a prescription. The package insert should be consulted for information about storage and USP controlled room temperature.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.