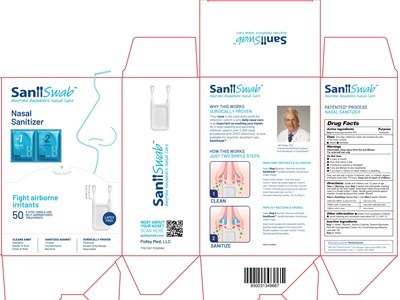
FDA Label for Saniiswab
View Indications, Usage & Precautions
Saniiswab Product Label
The following document was submitted to the FDA by the labeler of this product Ido Pharm. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Otc - Active Ingredient
isopropyl alcohol 70%
Inactive Ingredient
Water
Otc - Purpose
two-step method to clean and sanitize the skin of the inner nostrils
■ cleans ■ sanitizes
Otc - Keep Out Of Reach Of Children
Keep out of reach of children
Indications & Usage
Adults and children over 12 years of age
Step 1. Cleaning. Open Step 1 packet and hold handle, inserting
one swab tip into each nostril. Swab lower interior lining inside the
nose in a circular motion 7 times. Handle guard protects against
insertion of swab tip beyond the nostrils. Discard.
Step 2. Sanitizing. Repeat Step 1 with Step 2 packet. Discard
Warnings
Flammable, keep away from fire and flames
For external use only
Dosage & Administration
For external use only
Package Label.Principal Display Panel
* Please review the disclaimer below.