FDA Label for Bt100s
View Indications, Usage & Precautions
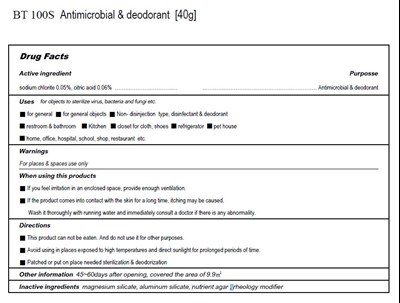
Bt100s Product Label
The following document was submitted to the FDA by the labeler of this product Sejin E&p Co., Ltd.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Otc - Active Ingredient
sodium chlorite 0.05%
citric acid 0.06%
Inactive Ingredient
magnesium silicate, aluminum silicate, nutrient agar, rheology modifier
Otc - Purpose
for objects to sterilize virus, vacteria and fungi etc.
Otc - Keep Out Of Reach Of Children
KEEP OUT OF REACH OF THE CHILDREN
Indications & Usage
This product can not be eaten. And do not use it for other purposes
Avoid using in places exposed to high temperatures and direct sunlight for prolonged periods of time.
Put or Inserted in employee ID case, handbag, wallet, pocket or mask storage case.
Warnings
If you feel irritation in an enclosed space, provide enough ventilation
If the product comes into contact with the skin for a long time, itching may be caused.
Wash it thoroughly with running water and immediately consult a doctor if there is any abnormality.
Dosage & Administration
For places & spaces use only
Package Label.Principal Display Panel
* Please review the disclaimer below.