Product Images Ticagrelor
View Photos of Packaging, Labels & Appearance
- 1 - ticagrelor tablets 1
- 9 - ticagrelor tablets 10
- 10 - ticagrelor tablets 11
- 11 - ticagrelor tablets 12
- 12 - ticagrelor tablets 13
- 13 - ticagrelor tablets 14
- 14 - ticagrelor tablets 15
- 15 - ticagrelor tablets 16
- 16 - ticagrelor tablets 17
- 17 - ticagrelor tablets 18
- 18 - ticagrelor tablets 19
- 2 - ticagrelor tablets 2
- 3 - ticagrelor tablets 3
- 4 - ticagrelor tablets 4
- abc - ticagrelor tablets 5
- 5 - ticagrelor tablets 6
- ticagrelor-tablets-60mg-60s - ticagrelor tablets 60mg 60s
- 6 - ticagrelor tablets 7
- 7 - ticagrelor tablets 8
- 8 - ticagrelor tablets 9
- ticagrelor-tablets-90mg-60s - ticagrelor tablets 90mg 60s
Product Label Images
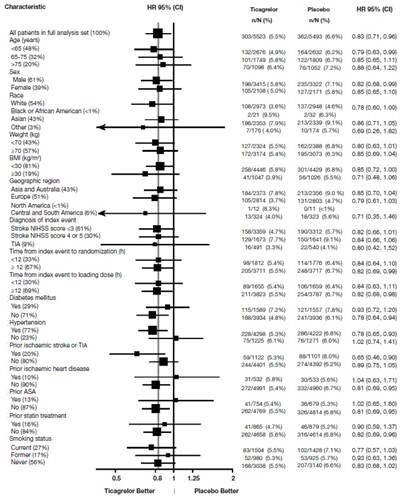
The following 21 images provide visual information about the product associated with Ticagrelor NDC 77771-522 by Radha Pharmaceuticals Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
9 - ticagrelor tablets 10

This text outlines recommendations for adjusting Ticagrelor doses when interacting with different drugs. It includes information on drugs such as Simvastatin, Atorvastatin, Levonorgestrel, Ethinyl Estradiol, Tolbutamide, Digoxin, and Cyclosporine. Specific dose adjustments and monitoring suggestions are provided based on the interacting drug to ensure safe and effective use of Ticagrelor.*
12 - ticagrelor tablets 13
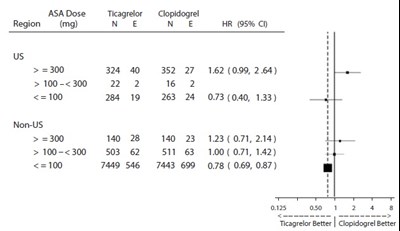
This text seems to be data related to a comparison between Ticagrelor and Clopidogrel dosages, categorizing regions and outcomes in terms of efficacy. It includes information on the number of participants, hazard ratios, and confidence intervals for different dosage ranges in US and non-US regions. The last line seems to suggest a comparison verdict stating "Ticagrelor Better" over "Clopidogrel Better."*
ticagrelor-tablets-60mg-60s - ticagrelor tablets 60mg 60s

Each tablet contains 60 mg ticagrelor. This medication is intended for adults - dosage requirements can be found in the package insert. Safety precautions should be observed, including keeping the medication out of children's reach. Storage temperature should not exceed 86°F. The Medication Guide can be accessed at the provided website. Dispense the Medication Guide alongside the medication to each patient. The medication is manufactured by ScieGen Pharmaceuticals, Inc. and distributed by Radha Pharmaceuticals, Inc.*
6 - ticagrelor tablets 7

This text provides information about IPA (%) induced by 20uM ADP over time with Ticagrelor, Clopidogrel, and Placebo. It seems to be related to a study or experiment tracking the effects of these substances on platelet aggregation.*
7 - ticagrelor tablets 8
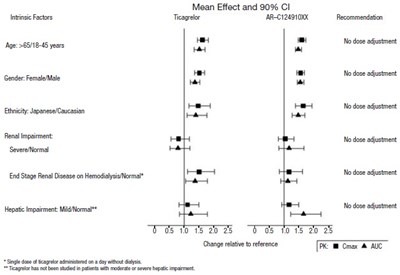
This text outlines the mean effect and 90% confidence intervals for the drug Ticagrelor ARI24910 along with recommendations for its use based on intrinsic factors like age, gender, ethnicity, renal function, and hepatic impairment. It specifies that no dose adjustments are needed for individuals falling into particular categories within these factors. Additionally, it notes that Ticagrelor can be administered in patients with moderate to severe hepatic impairment.*
8 - ticagrelor tablets 9

This is a list of interacting drugs and their effects on Ticagrelor AR-C124910XX with recommendations based on the strength of interactions, such as strong, moderate, or potent inhibitors and inducers of CYP3A4. The text provides specific examples of medications like Ketoconazole, Diltiazem, Rifampin, Aspirin, Desmopressin, Heparin, and Enoxaparin, along with their recommended dosages and administration frequency. Additionally, it mentions P-glycoprotein inhibitors like Cyclosporine and Fentanyl with their respective doses. The document also includes changes relative to a reference point, potentially regarding pharmacokinetics.*
ticagrelor-tablets-90mg-60s - ticagrelor tablets 90mg 60s

This text provides information about Ticagrelor tablets, with each tablet containing 90 mg of Ticagrelor. The usual adult dosage should be confirmed by referring to the package insert. It includes a warning to keep the medication out of the reach of children and to store it at 25°C (77°F). The text also mentions a link to print the Medication Guide and provides details about the manufacturer and distributor.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.