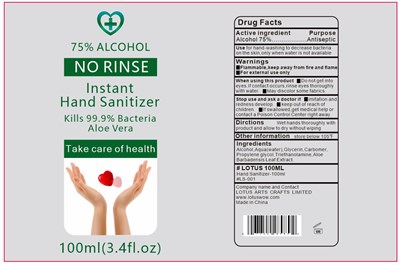
FDA Label for Instant Hand Sanitizer
View Indications, Usage & Precautions
Instant Hand Sanitizer Product Label
The following document was submitted to the FDA by the labeler of this product Lotus Arts Crafts Limited. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient(S)
Alcohol 75%
Purpose
Antiseptic
Use For Hand
washing to decrease bacteria on the skin ,only when water is not available
Warnings
For external use only. Flammable. Keep away from heat or flame
Do Not Use
Do not get into eyes. If contact occurs ,rinse eyes thoroughly with water
Otc - When Using
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Otc - Stop Use
Imitation and redness develop.
Keep out of reach of children.
If swallowed ,get medical help for contact a Poison Control Center right away.
Otc - Keep Out Of Reach Of Children
Keep out of reach of children.
Directions
Wet hands thoroughly with product and allow to dry without wiping
Other Information
Store below 105° F
Inactive Ingredients
Aqua(water) ,Glycerin, Carbomer, Propylene glycol, Triethanolamine ,Aloe Barbadensis Leaf Extract
Package Label - Principal Display Panel
* Please review the disclaimer below.