Product Images Livmarli
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 29 images provide visual information about the product associated with Livmarli NDC 79378-110 by Mirum Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure A - livmarli 02
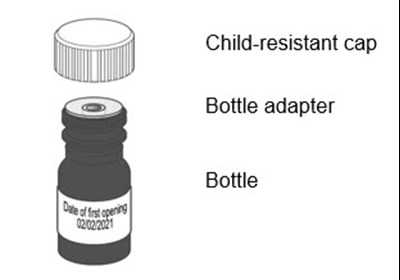
This product appears to include a child-resistant cap and a bottle adapter designed to fit onto a bottle. No further information is available to provide a more detailed description or context.*
Figure G - livmarli 08
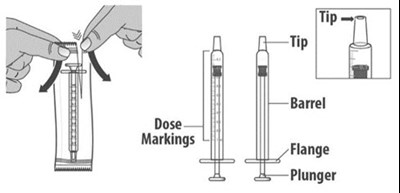
This is information about a medical device, likely a syringe. It includes markings for the dose and the tip of the device, as well as information about the barrel, flange, and plunger. There is an "E-S" and a "5" marked on the device.*
livmarli 29

This is a description of a medication with the National Drug Code (NDC) 79378-110-01. The medication is called N Llvmarllm and is recommended to be stored unopened at 20°C - 25°C (68°F - 77°F). After opening, it should be stored at the same temperature range and should be disposed of any remaining medication after 45 days. The medication should be kept out of the reach of children. The dosage information is provided in the prescribing information. The medicine has been manufactured by Mirum Pharmaceuticals, Inc. in Foster City, California, and is made in Canada. The LOT is mentioned but the XXXXX is not identifiable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

























