Product Images Quviviq
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Quviviq NDC 80491-7825 by Idorsia Pharmaceuticals Ltd, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - quviviq 02
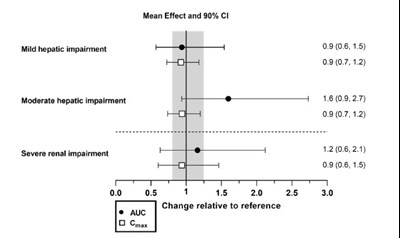
This appears to be a table of statistical data representing mean effects and 90% confidence intervals for different levels of hepatic and renal impairment. There is also information about relative changes. However, without additional context, it's not clear what these numbers represent or what they are being used for.*
Figure 2 - quviviq 03

This is a table showing the mean effect and 90% confidence interval for different compounds interacting with CYP3A4 inhibitors, gastric pH modifiers, and an SSRI. The table includes the names of the compounds and their respective mean effect values and confidence interval ranges. The compounds include Ranitidine, Diltiazem, Efavirenz, Famotidine, Aol ol, and Galopram. The predicted value is shown for Ranitidine.*
Figure 4 - quviviq 05
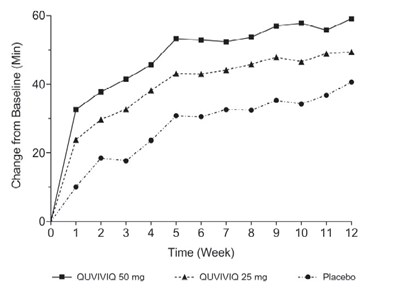
This is a chart showing changes from baseline (minimum) over time in patients on QUVIVIQ 50 mg, QUVIVIQ 25 mg, and Placebo. The X-axis represents the time in weeks, while the Y-axis represents the change in baseline in minutes. There is no further information available.*
PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Carton - quviviq 06

Daridorexant is a medication available in the form of 25 mg tablets. Each tablet comprises daridorexant hydrochloride of 27 mg. The recommended storage temperature is between 20°C to 25°C, and excursions are allowed between 15°C to 30°C. The medication guide should be delivered to each patient at the time of dispensing. The drug is produced in Austria and being distributed in the United States by dorsia Pharmaceuticals US Inc. This medication is only available with a prescription.*
PRINCIPAL DISPLAY PANEL - 50 mg Tablet Bottle Carton - quviviq 07

"Quvivia™ 1" is a tablet containing daridorexant, specifically daridorexant hydrochloride, in 50 mg dosage. This medication is for oral use only and must be kept out of reach of children. It should be stored between 20°C to 25°C or between 15°C to 30°C, with excursions permitted. It is distributed by Idorsia Pharmaceuticals US Inc and there are 130 tablets in a package. The full prescribing information can be found in the package insert."*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

