Product Images Timolol Maleate
View Photos of Packaging, Labels & Appearance
- structure - b666fc6c b409 45a6 a890 fa5b41585390 01
- figure a - b666fc6c b409 45a6 a890 fa5b41585390 02
- figure b - b666fc6c b409 45a6 a890 fa5b41585390 03
- fugure c - b666fc6c b409 45a6 a890 fa5b41585390 04
- 0.25per5mllabel - b666fc6c b409 45a6 a890 fa5b41585390 05
- 0.25per5mlcarton - b666fc6c b409 45a6 a890 fa5b41585390 06
- 0.25per10mllabel - b666fc6c b409 45a6 a890 fa5b41585390 07
- 0.25per10mlcarton - b666fc6c b409 45a6 a890 fa5b41585390 08
- 0.5per5mllabel - b666fc6c b409 45a6 a890 fa5b41585390 09
- 0.5per5mlcarton - b666fc6c b409 45a6 a890 fa5b41585390 10
- 0.5per10mllabel - b666fc6c b409 45a6 a890 fa5b41585390 11
- 0.5per10mlcarton - b666fc6c b409 45a6 a890 fa5b41585390 12
Product Label Images
The following 12 images provide visual information about the product associated with Timolol Maleate NDC 81469-210 by First Nation Group, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
0.25per5mllabel - b666fc6c b409 45a6 a890 fa5b41585390 05

This is information about Timolol Maleate Ophthalmic Solution, with a recommended usual adult dosage of 1 drop in the eye once or twice daily. It should be stored between 20°C to 25°C and protected from freezing and light. The solution can be used until the expiration date on the bottle. This product is manufactured in Germany and is for topical application in the eye. It is a sterile solution, and each mL contains timolol maleate equivalent to 3.4 mg/mL, equal to 2.5 mg/mL of timolol.*
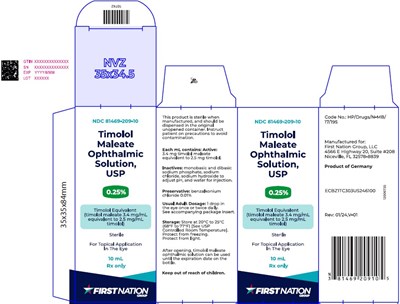
0.25per5mlcarton - b666fc6c b409 45a6 a890 fa5b41585390 06
This is a description for Timolol Maleate Ophthalmic Solution. The product comes in a sterile 35x35x84mm bottle with NDC 81469-209-05. Each mL of solution contains Timolol Maleate equivalent to 0.25 mg Timolol. It is for topical application in the eye. The product should be stored at controlled room temperature and protected from light. Manufactured for First Nation Group, LLC in Germany. Keep out of reach of children. Rx only.*
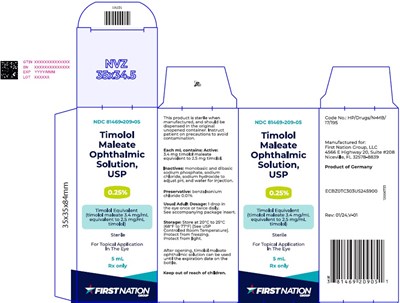
0.25per10mllabel - b666fc6c b409 45a6 a890 fa5b41585390 07

This is a description of an ophthalmic solution containing Timolol Maleate. It is used for topical application in the eye and has a concentration of timolol maleate. The recommended dosage for adults is 1 drop in the eye once or twice daily. The solution should be stored at room temperature and protected from freezing and light. The solution can be used until the expiration date on the bottle after opening. It is manufactured for First Nation Group, LLC and is a product of Germany.*
0.5per5mllabel - b666fc6c b409 45a6 a890 fa5b41585390 09

This is a description of Timolol Maleate ophthalmic solution in a 5mL sterile bottle with a concentration of 0.5%. The equivalent of Timolol is 6.8 mg/mL, which is equivalent to 5 mg/mL of Timolol. It is recommended for topical application in the eye and the usual adult dosage is one drop in the eye once or twice daily. The solution should be protected from freezing and light, and can be used until the expiration date on the bottle. This product is manufactured for First Nation Group, LLC in Germany.*
0.5per5mlcarton - b666fc6c b409 45a6 a890 fa5b41585390 10

This is a description of Timolol Maleate Ophthalmic Solution. It comes in a sterile solution format for topical application to the eye. Each milliliter contains 5% of active ingredient Timolol Maleate. It includes preservatives and inactive ingredients. Storage instructions recommend keeping it between 20°C to 25°C. The product is manufactured in Germany and distributed by First Nation Group, LLC. It is essential to follow the provided dosage instructions and keep it out of children's reach.*
0.5per10mllabel - b666fc6c b409 45a6 a890 fa5b41585390 11

This text provides dosage and storage information for Timolol Maleate ophthalmic solution. The usual adult dosage is 1 drop in the eye once or twice daily. It should be stored at 20°C to 25°C. After opening, the solution can be used until the expiration date on the bottle. The solution should be protected from freezing and light. The product is manufactured in Germany and has an expiration date specified on the bottle.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.