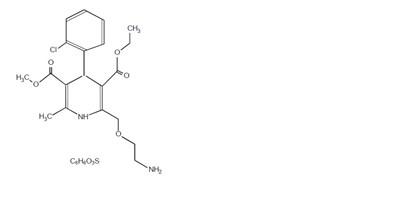
Product Images Amlodipine Besylate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Amlodipine Besylate NDC 82009-026 by Quallent Pharmaceuticals Health Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image2 - c45b7a8d 29fb 44b3 929d 8c6e2a41ff11 02

This appears to be a section of a medical or scientific report discussing an event rate and related statistics. The P-value is 0.003, the hazard ratio is 0.691, and the 95% confidence interval ranges from 0.54 to 0.88. There is also a chart with time in months and a reference to Amlodipine, a medication used to treat high blood pressure. However, the context and purpose of the study are not clear from this limited text.*
Image3 - c45b7a8d 29fb 44b3 929d 8c6e2a41ff11 03

This appears to be a table containing statistical data related to a medical study involving a drug called Amlodipine and a placebo. The data is organized by age, gender, blood pressure, vessel disease, and stent placement. The table includes the number of participants in each group and various measurements such as mean sitting systolic blood pressure. Additional information is not available.*
1 - c45b7a8d 29fb 44b3 929d 8c6e2a41ff11 04

Each tablet contains Amlodipine besylate USP equivalent, manufactured by Cipla Ltd. The tablets are dispensed in tight, light-resistant containers, and should be stored in a controlled room temperature. The usual dosage should be seen in the accompanying prescribing information. Please refer to the Patient Leaflet for further instructions. Batch overprinting includes Serial No., Expiry & Lot. PHARMACIST: ExP will be overprinted during commercial packing.*
2 - c45b7a8d 29fb 44b3 929d 8c6e2a41ff11 05

Each tablet contains Amlodipine besylate USP 5 mg. It is manufactured by Cpla Ltd in Verna Goa, India and sold under the brand name Quallent. The usual dosage is specified in the accompanying prescribing information. The tablets should be stored in a tight, light-resistant container at room temperature and protected from light and moisture. The patient leaflet should be dispensed with the medicine. Batch details including serial number, expiry, and lot numbers will be overprinted during commercial packing.*
3 - c45b7a8d 29fb 44b3 929d 8c6e2a41ff11 06
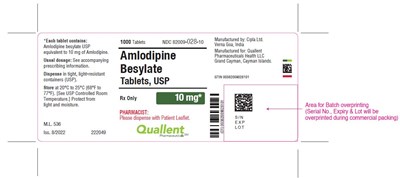
This appears to be the description of a medication named Amlodipine Besylate, in the form of 1000 tablets per container, with a concentration of 1 mg. The manufacturer is Cila Ltd. and the product is being manufactured for Qualent. The text also provides the National Drug Code (NDC) of 62000-028-10, and some instructions for storage and dispensing of the medication. There is a reference to "accompanying Grand Cayman, Cayman Islands prescribing information," but it is unclear what this refers to.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.